- Title
-
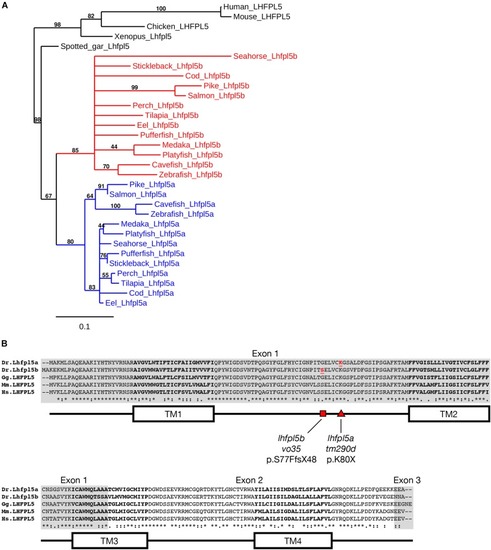
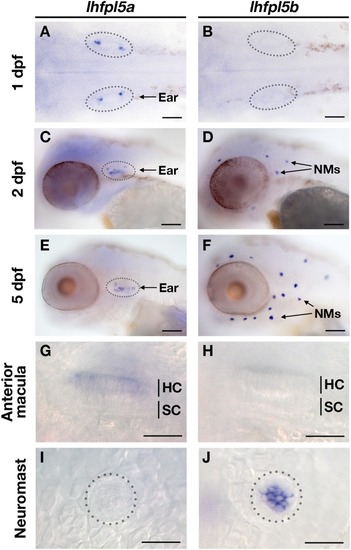
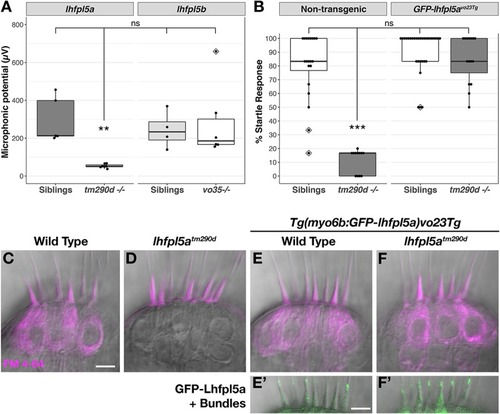
The lhfpl5 Ohnologs lhfpl5a and lhfpl5b Are Required for Mechanotransduction in Distinct Populations of Sensory Hair Cells in Zebrafish
- Authors
- Erickson, T., Pacentine, I.V., Venuto, A., Clemens, R., Nicolson, T.
- Source
- Full text @ Front. Mol. Neurosci.
|
Duplicated |
|
Zebrafish |
|
|
|
|
|
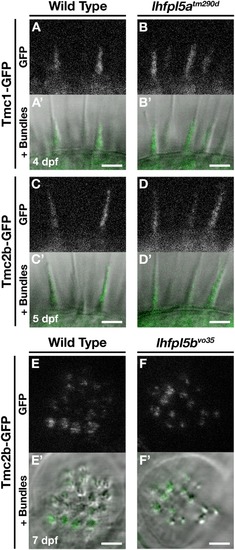
Tmc proteins do not require Lhfpl5a or Lhfpl5b for localization to the stereocilia of zebrafish hair cells. |
|
Lhfpl5a requires MET complex proteins Pcdh15a, Cdh23, and Myo7a for normal localization in the stereocilia of zebrafish hair cells. EXPRESSION / LABELING:
PHENOTYPE:
|