- Title
-
Identification and characterization of T reg-like cells in zebrafish
- Authors
- Kasheta, M., Painter, C.A., Moore, F.E., Lobbardi, R., Bryll, A., Freiman, E., Stachura, D., Rogers, A.B., Houvras, Y., Langenau, D.M., Ceol, C.J.
- Source
- Full text @ J. Exp. Med.

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. EXPRESSION / LABELING:
|
|
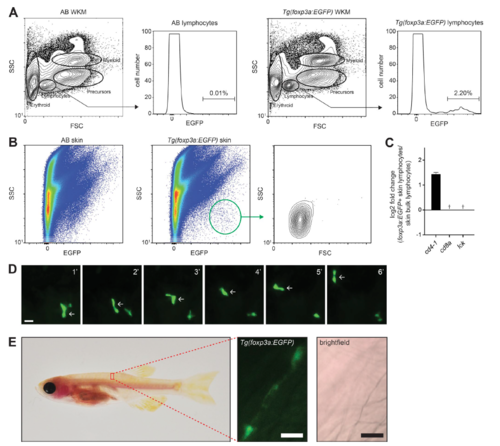
Identification and analysis of foxp3a:EGFP-positive thymocytes. (A) Flow cytometry analysis of thymus from representative AB and Tg(foxp3a:EGFP) animals. (B) qRT-PCR of selected genes in foxp3a:EGFP-positive thymocytes relative to bulk thymocytes. Relative CD4 and CD8 expression is also shown and represents zebrafish cd4-1 and cd8a genes. †, Expression of cd8a was below the limit of detection in foxp3a:EGFP-positive thymocytes. Error bars indicate SEM.; n = 3. (C) Cells from dissected thymus of a Tg(foxp3a:EGFP); Tg(rag2:mCherry) animal showing a double-positive thymocyte (arrowheads). Bars, 10 µm. EXPRESSION / LABELING:
|

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. |
|
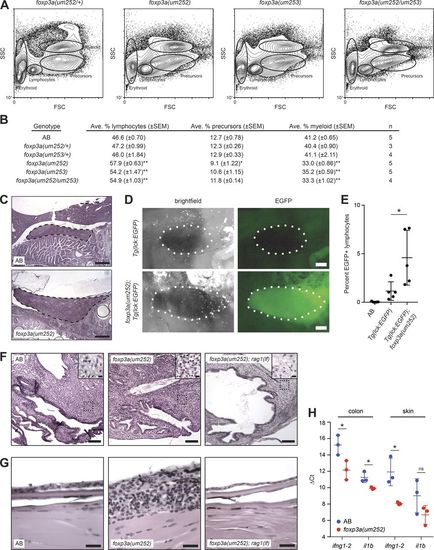
Lymphocyte expansion and lymphocyte-dependent inflammation in foxp3a mutants. (A) Flow cytometry analyses of WKM from representative foxp3a mutant animals. (B) Differential counts of nonerythroid cells from WKM. Counts were obtained from at least 200 cells per WKM cytospin with May-Grünwald Giemsa staining. Two-tailed Student’s t test (foxp3a mutant vs. AB), *, P < 0.05; **, P < 0.001. (C) Representative images of hematoxylin and eosin–stained sagittal sections of spleens from wild-type AB and foxp3a(um252) mutants. Images showing largest extent of splenic size are shown. Spleens are shown bordered by dashed lines. Bars, 250 µm. (D) Representative images of dissected spleens from Tg(lck:EGFP) and Tg(lck:EGFP); foxp3a(um252) animals. Spleens are shown bordered by dotted lines. Bars, 250 µm. (E) Flow cytometry analysis of spleens from AB (n = 5), Tg(lck:EGFP) (n = 5) and Tg(lck:EGFP); foxp3a(um252) (n = 5) animals. Percentages of EGFP-positive cells in the lymphocyte gate are shown. Two-tailed Student’s t test, *, P < 0.05. (F) Representative images of hematoxylin and eosin–stained sagittal sections of urogenital pores from wild-type AB, foxp3a(um252), and foxp3a(um252); rag1(lf) animals. In total, 0/4 wild-type AB, 6/6 foxp3a(um252), and 0/3 foxp3a(um252); rag1(lf) animals displayed connective tissue inflammation. Insets, increased cellularity characterized by marked influx of lymphocyte-predominant inflammatory cells (arrowheads) in foxp3a(um252) mutants compared with the wild-type AB and foxp3a(um252); rag1(lf) double mutants. Bars: 100 µm; (inset) 10 µm. (G) Representative images of hematoxylin and eosin–stained sagittal sections of skin from wild-type AB, foxp3a(um252), and foxp3a(um252); rag1(lf) animals. In total, 0/4 wild-type AB, 5/6 foxp3a(um252), and 0/3 foxp3a(um252); rag1(lf) animals displayed skin inflammation. Bar, 25 µm. (H) qRT-PCR analysis of inflammation marker genes ifng1-2 and il1b. ΔCt values were calculated relative to a β-actin control. Two-tailed Student’s t test, *, P < 0.05; ns, not significant. Error bar indicates SEM; n = 3. |
|
Lymphocyte expansion and lymphocyte-dependent inflammation in foxp3a mutants. (A) Flow cytometry analyses of WKM from representative foxp3a mutant animals. (B) Differential counts of nonerythroid cells from WKM. Counts were obtained from at least 200 cells per WKM cytospin with May-Grünwald Giemsa staining. Two-tailed Student’s t test (foxp3a mutant vs. AB), *, P < 0.05; **, P < 0.001. (C) Representative images of hematoxylin and eosin–stained sagittal sections of spleens from wild-type AB and foxp3a(um252) mutants. Images showing largest extent of splenic size are shown. Spleens are shown bordered by dashed lines. Bars, 250 µm. (D) Representative images of dissected spleens from Tg(lck:EGFP) and Tg(lck:EGFP); foxp3a(um252) animals. Spleens are shown bordered by dotted lines. Bars, 250 µm. (E) Flow cytometry analysis of spleens from AB (n = 5), Tg(lck:EGFP) (n = 5) and Tg(lck:EGFP); foxp3a(um252) (n = 5) animals. Percentages of EGFP-positive cells in the lymphocyte gate are shown. Two-tailed Student’s t test, *, P < 0.05. (F) Representative images of hematoxylin and eosin–stained sagittal sections of urogenital pores from wild-type AB, foxp3a(um252), and foxp3a(um252); rag1(lf) animals. In total, 0/4 wild-type AB, 6/6 foxp3a(um252), and 0/3 foxp3a(um252); rag1(lf) animals displayed connective tissue inflammation. Insets, increased cellularity characterized by marked influx of lymphocyte-predominant inflammatory cells (arrowheads) in foxp3a(um252) mutants compared with the wild-type AB and foxp3a(um252); rag1(lf) double mutants. Bars: 100 µm; (inset) 10 µm. (G) Representative images of hematoxylin and eosin–stained sagittal sections of skin from wild-type AB, foxp3a(um252), and foxp3a(um252); rag1(lf) animals. In total, 0/4 wild-type AB, 5/6 foxp3a(um252), and 0/3 foxp3a(um252); rag1(lf) animals displayed skin inflammation. Bar, 25 µm. (H) qRT-PCR analysis of inflammation marker genes ifng1-2 and il1b. ΔCt values were calculated relative to a β-actin control. Two-tailed Student’s t test, *, P < 0.05; ns, not significant. Error bar indicates SEM; n = 3. EXPRESSION / LABELING:
|
|
Phenotypic analyses of foxp3a mutants. (A) Flow cytometry analysis of WKM from a representative Tg(foxp3a :EGFP); foxp3a(um252) animal. Gated lymphocytes are plotted as a histogram with the percentage of EGFP-positive lymphocytes indicated. (B) Percentages of foxp3a :EGFP -positive lymphocytes, based on flow cytometry analysis, are increased in a foxp3a(um252) mutant (n = 5) compared with a wild-type (n = 5) background. Two-tailed Student’s t test, *, P < 0.05. Error bar indicates SEM. (C) qRT-PCR of indicated genes in EGFP-positive lymphocytes sorted from Tg(foxp3a :EGFP); foxp3a(um252) and Tg(foxp3a :EGFP) animals. The log2 fold change of gene expression in EGFP-positive lymphocytes from Tg(foxp3a :EGFP); foxp3a(um252) animals compared with EGFP-positive lymphocytes from Tg(foxp3a :EGFP) animals is plotted. †, Expression of foxp3b was below the limit of detection in EGFP-positive lymphocytes from both Tg(foxp3a :EGFP); foxp3a(um252) and Tg(foxp3a :EGFP) animals. Error bar indicates SEM; n = 3. (D) Images of representative sibling foxp3a(um252)/+ heterozygotes and foxp3a(um252) homozygotes. (E) Normalized spleen sizes of sibling foxp3a(um252/+) (n = 4) and foxp3a(um252/um252) (n = 6) animals. Spleen sizes (μm2 ×103/mm3) were estimated by normalizing maximum spleen area (area at level of splenic artery) by fish volume. Two-tailed Student’s t test, *, P = 0.007. (F) Flow cytometry analysis of a representative Tg(lck :EGFP) spleen. The lymphocyte-containing gate is indicated. (G) Gated lymphocytes from representative animals are plotted to indicate EGFP positivity. (H) qRT-PCR analysis of inflammation marker gene tnfa. ΔCt values were calculated relative to a β-actin control. Two-tailed Student’s t test, ns, not significant. Error bar indicates SEM; n = 3. PHENOTYPE:
|