- Title
-
A novel brain tumour model in zebrafish reveals the role of YAP activation in MAPK/PI3K induced malignant growth
- Authors
- Mayrhofer, M., Gourain, V., Reischl, M., Affaticati, P., Jenett, A., Joly, J.S., Benelli, M., Demichelis, F., Poliani, P.L., Sieger, D., Mione, M.
- Source
- Full text @ Dis. Model. Mech.
|
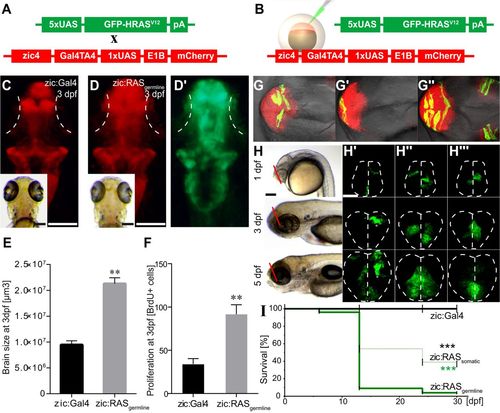
Oncogenic RAS induces proliferation of neural progenitor cells, clonal expansion and reduced survival. (A,B) Schematic representation of tumour induction through (A) the cross between zebrafish lines carrying the indicated transgenes or (B) the injection of the oncogenic construct (green) into zic:Gal4 embryos to express UAS:GFP-HRASV12 specifically in the brain. (C,D) Dorsal view of representative 3 dpf images of larvae showing the telencephalon in a control larva (C, zic:Gal4, mCherry expression) compared with that of an oncogenic larva (D, zic:RASgermline, mCherry expression; D′, zic:RASgermline, UAS:GFP-HRASV12 expression), white dotted lines mark the eyes. (E) Quantification of brain size reveals a doubling in size of the zic:RASgermline-expressing tissue. (F) Counting of BrdU-positive cells in the telencephalon of 3 dpf larvae reveals doubling in the number of proliferating cells in zic:RASgermline versus zic:Gal4 controls. (G-G″) Dorsal view of three 1 dpf zic:RASsomatic larvae showing individual clones expressing UAS:GFP-HRASV12. (H-H″′) Lateral bright-field and coronal confocal images of the telencephalon of three live zic:RASsomatic larvae (plane of focus indicated by red lines in H) at 1 dpf, 3 dpf and 5 dpf revealing clonal expansion of oncogene-expressing cells in the same larvae from 1 to 5 dpf. White dotted lines mark the outline of the brain. (I) Survival curve of zic:RASsomatic larvae (green dashed line; n=166) compared with zic:Gal4 controls (black line, black asterisk; n =105) and zic:RASgermline larvae (green solid line, green asterisk; n =255). Data are represented as mean±s.d. **P<0.03, ***P<0.001. Scale bars: 500 µm in C,D; 500 µm in H; 50 µm in H′. EXPRESSION / LABELING:
PHENOTYPE:
|
|
Somatic expression of oncogenic RAS induces tumour development and heterotopia. (A-C) Fluorescence images of 2 months post-fertilisation (mpf) zebrafish brains of zic:Gal4 (A) and zic:RASsomatic (B,C) fish showing (A) a control brain, (B) tumours in the telencephalon (T), diencephalon (D) and IVth ventricle region (V), and (C) heterotopia (H). (D) Graph representing the frequency of different lesions resulting from induction of UAS:GFP-HRASV12 expression (n=134, of which T=83, D=40, V=44, H=67). (E-G) Volume rendering of 3D reconstructions of two brains with tumours (E,F, green areas) and a brain with heterotopia (G, white arrow) shown as dorsal view (inset) and one sagittal section (large image). (H) 3D reconstruction and volume rendering of a brain showing different tumour expansions (colour coded according to histological and anatomical features and GFP expression). Scale bars: 2 mm in A-C. |
|
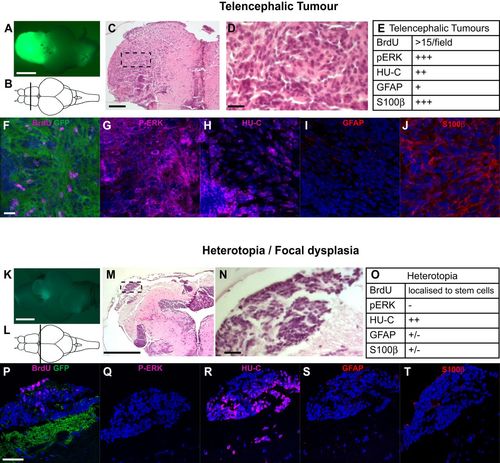
Histological and immunological appearance of telencephalic tumour and heterotopia. (A) Representative telencephalic tumour in zic:RASsomatic fish. (B) Schematic drawing, indicating the position of the sections shown in F-J. (C) H&E-stained section, boxed area indicates enlargement shown in D. (E) Summary of the immunohistochemical observations related to telencephalic tumours. (F-J) Immunostaining of telencephalic tumour sections stained as indicated. DAPI as counterstaining is in blue. (K) Representative heterotopia in zic:RASsomatic fish. (L) Schematic drawing, indicating the position of the sections shown in P-T. (M) H&E-stained section, boxed area indicates enlargement shown in N. (O) Summary of the immunohistochemical observations related to heterotopia. (P-T) Immunostaining of telencephalic tumour sections stained as indicated. DAPI as counterstaining is in blue. Scale bars: 2 mm in A,K; 200 µm in C,M; 25 µm in D,F-J,N,P-T. |

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. PHENOTYPE:
|
|
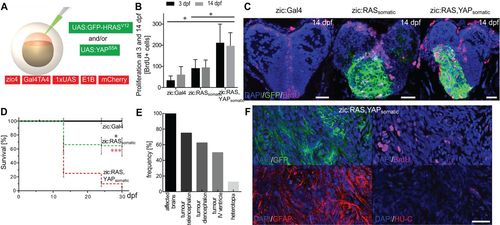
YAP activation promotes tumour growth. (A) Schematic representation of tumour induction through the co-injection of oncogenic constructs (green) into zic:Gal4 embryos. (B) Quantification of BrdU-positive cells in the telencephalon of 3 and 14 dpf juveniles reveals doubling in the number of proliferating cells in zic:RAS,YAPsomatic fish compared with zic:RASsomatic fish and zic:Gal4 control fish. (C) Confocal image of 14 dpf fish showing BrdU expression (magenta) in zic:Gal4, zic:RASsomatic and zic:RAS,YAPsomatic. (D) Survival rate of zic:RASsomatic (green dashed line; n=166) compared with zic:Gal4 (black solid line, black asterisk; n=105) and zic:RAS,YAPsomatic (red dashed line, red asterisks; n=100). (E) Graph representing the frequency of different lesions resulting from zic:RAS,YAPsomatic expression as determined by stereomicroscopic analysis with tumours in telencephalon (T), diencephalon (D) and IVth ventricle (V), and heterotopia (H). (F) Immunostaining for GFP, BrdU, GFAP and HU-C of zic:RAS,YAPsomatic fish. Data are represented as mean±s.d. *P<0.05; ***P<0.001. Scale bars: 20 µm in C,F. |
|
Gal4 expression pattern in control fish (zic:Gal4). A-C) Visualisation of zic:Gal4 expression during zebrafish development as indicated. Bright field images (A-C) and Gal4 expression (A'-C') visualised through mCherry fluorescence at 2 dpf (A), 5 dpf (B) and in the brain at 2 mpf (C). Scale bars: A-B = 500 μm, C = 2 mm. EXPRESSION / LABELING:
|
|
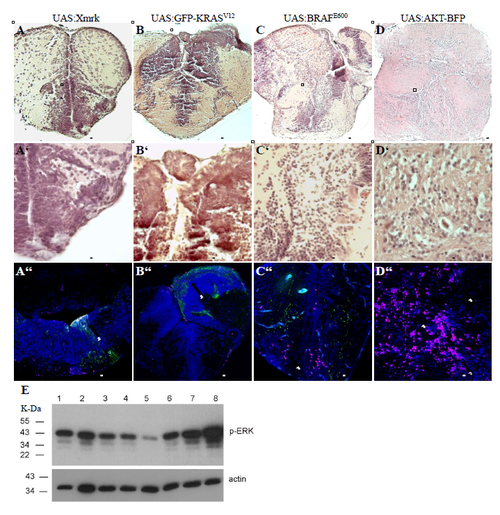
Tumours can be induced by different oncogenes. A-D) H&E staining of lesions induced by somatic expression of different oncogenes. Boxed area indicates enlargement shown in A'-D', respectively. A''-D'') Immunostaining for BrdU of lesions induced by somatic expression of different oncogenes. Arrows point at lesions. E) Western blot showing the levels of phosphorylated ERK (P-ERK) and actin in control brain (lane 1), heterotopia (lane 2), zic:EGFRVIIIsomatic (lane 3), zic:Xmrksomatic (lane 4), zic:AKT-BFPsomatic (lane 5), zic:BRAFME600 somatic (lane 6), zic:GFP-KRASV12 somatic (lane 7) and zic:GFP-HRASV12 somatic (lane 8). Scale bars: A-C = 100 μm; D = 1 mm; A'-D' = 50 μm; A''-D'' = 100 μm. |
|
Histological and immunological appearance of IVth ventricle and diencephalic tumours. A) Representative diencephalic tumour in zic:RASsomatic fish. B) Schematic drawing, indicating the position of the sections shown in F-J. C) H&E stained section, boxed area indicates enlargement shown in D. E) Summary of the immunohistochemical observations related to diencephalic tumours. F-J) Immunostaining of heterotopia sections stained as indicated. K) Representative IVth ventricle tumour in zic:RASsomatic fish. L) Schematic drawing, indicating the position of the sections shown in P-T. M) H&E stained section, boxed area indicates enlargement shown in N. O) Summary of the immunohistochemical observations related to IV ventricle tumours. P-T) Immunostaining of IVth ventricle tumour sections stained as indicated. Scale bars: A, K= 2 mm; C, M=200 μm; D, F-J, N, PT = 25 μm. |
|
Heterotopia occurs also in telencephalon and cerebellum. A) Representative telencephalic heterotopia in zic:RASsomatic, boxed area indicates enlargement shown in A'. B) Representative IVth ventricle heterotopia zic:RASsomatic, boxed area indicates enlargement shown in B'. Black arrows point at abnormally localised neural cells (heterotopia). Scale bars: A, B = 100 μm; A', B' = 20 μm. |
|
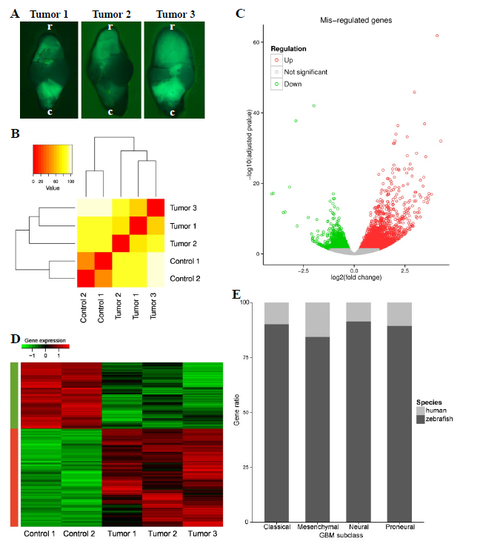
Quality control for zebrafish model RNAseq results. A) Dorsal view of stereo images of whole brains from zic:RASsomatic used for RNAseq analysis (r=rostral, c=caudal). B) Hierarchical clustering of control and tumour samples using Euclidean distance on gene expression grouped the samples according to tumour or control. C) The volcano plot represents all known zebrafish genes for which a differential expression analysis was done. The significantly differentially expressed (DE) genes (adjusted p‑value<0.05) are coloured in red (up-regulated) or green (down-regulated). D) After clustering of significantly (adjusted p-value<0.05) DE genes based on normalised expression, the genes clustered according to regulation, verifying the efficiency of the normalisation step. E) After the identification of the zebrafish orthologs of the 840 GBM human markers, through Ensembl annotation and manual refinement, more than 80 % of genes could be identified for all four GBM subclasses. |
|
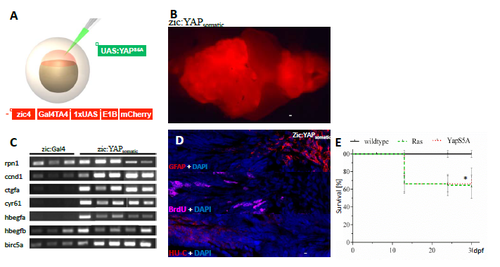
Ectopic YAP expression induces brain tumour development. A) Schematic representation of tumour induction through the injection of oncogenic constructs (green) into zic:Gal4 embryos. B) Stereo image of a tumour developed in zic:YAPsomatic. C) YAP target gene expression in zic:Gal4 control brains and zic:YAPsomatic tumours. D) Immunostaining for BrdU, GFAP and HU-C of zic:YAPsomatic tumours. E) Survival rate of zic:RASsomatic (green dashed line) and zic:YAPsomatic (red dotted line) compared to zic:Gal4 (black solid line, black asterix). Data are represented as mean ± SD. Scale bars: B = 2 mm; G = 20 μm; *p-value<0.05. |