- Title
-
Cyp2aa9 regulates haematopoietic stem cell development in zebrafish
- Authors
- Chen, J., He, J., Li, L., Yang, D., Luo, L.
- Source
- Full text @ Sci. Rep.
|
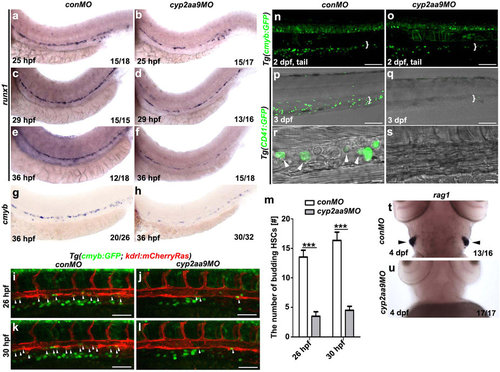
cyp2aa9 is required for the HSCs formation. (a-f) In the cyp2aa9 morphants, the expression of the definitive HSC precursors marker runx1 became decreased in the ventral floor of the dorsal aorta from 25 hpf to 36 hpf. (g,h) The number of cmyb-expressing HSCs was significantly reduced in cyp2aa9 morphants at 36 hpf. (i-l) In contrast to the control embryos at 26 hpf (i, n = 22/24) and 30 hpf (k, n = 23/24), the number of HSCs undergoing budding process from VDA (arrowheads) dramatically reduced in the cyp2aa9 morphant (j, n = 21/25; l, n = 21/25; Scale bar, 50 µm). (m) Quantifications of the number of HSCs undergoing budding process (n = 8, mean ± SD, ***p < 0.001, Student’s t-test). (n,o) In CHT (white brackets), the number of cmyb:GFP-positive cells was significantly reduced in cyp2aa9 morphants (27.6 ± 5.3, mean ± s.e.m., n = 6) comparing with the control (58.2 ± 4.1, mean ± s.e.m., n = 6). Scale bar, 100 µm. (p-s) Fluorecently labelled HSCs and DIC image showed the round-shaped HSCs (white arrowheads) were hardly detected in the CHT (white brackets) of cyp2aa9 morphants under the Tg(CD41:GFP) background (p, n = 24/24; q, n = 25/29; scale bar, 100 µm). CHT regions were zoomed in and showed in the (r,s) (scale bar, 10 µm). (t,u) The expression of T-lymphocyte marker rag1 in the thymus (arrowheads) dramatically decreased in the cyp2aa9 morphants. (a-s) Lateral views, anterior left, dorsal up. (t,u) Ventral views, anterior up. |
|
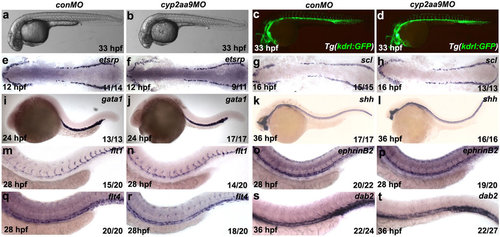
The non-HSC tissues are unaffected in the cyp2aa9 morphants. (a-d) The cyp2aa9 morphants in bright-field were morphologically normal (a, n = 110/110; b, n = 125/130) and with intact vasculature at 33 hpf under the Tg(kdrl:GFP) background (c, n = 21/21; d, n = 21/25). (e-t) The cyp2aa9 morphants displayed normal expressions of the following tissue-specific markers: primitive haematopoiesis (etsrp, e,f; scl, g,h; gata1, i,j), notochord (shh, k,l), dorsal aorta (flt1, m,n; ephrinB2, o,p) and posterior cardinal vein (flt4, q,r; dab2, s,t). (a-d,i-t) Lateral views, anterior left, dorsal up. (e-h) Dorsal views, anterior left. EXPRESSION / LABELING:
PHENOTYPE:
|
|
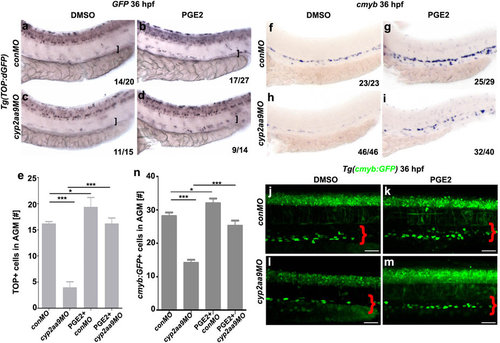
PGE2 rescues defective HSC formation and reduced Wnt/β-catenin activity caused by cyp2aa9MO. (a-d) Down-regulated Wnt/β-catenin activity in the VDA (brackets) caused by cyp2aa9MO was rescued by PGE2 as shown by WISH of GFP in the Tg(TOP:dGFP) Wnt-reporter line at 36 hpf. (e) Quantifications of total TOP-positive cells in the major trunk vessels (n = 6, mean ± SD). (f-i) Defective HSC formation in the cyp2aa9 morphants was rescued by PGE2 as shown by cmyb expression. (j-m) Decrease in the number of HSCs in the cyp2aa9 morphants was rescued by PGE2 as shown by the cmyb:GFP transgene. Scale bar, 50 µm. (n) Quantifications of the cmyb:GFP-positive cells in the AGM (n = 8, mean ± SD). *p < 0.05, ***p < 0.001, Student’s t-test. All the views of embryos are lateral, anterior left, dorsal top. |
|
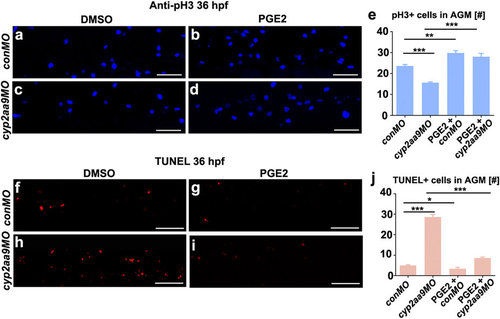
Defective proliferation and apoptosis caused by cyp2aa9MO were rescued by PGE2. (a-d) cyp2aa9MO led to reduced number of mitotic active cells at 36 hpf in the AGM as shown by pH3 antibody staining, which could be rescued by PGE2. (e) Quantification of pH3-positive cells in the AGM (n = 6, mean ± SD). (f-i) cyp2aa9MO led to increases in the number of apoptotic cells at 36 hpf in the AGM as shown by TUNEL assay, which could be rescued by PGE2. (j) Quantification of the number of apoptotic cells in the AGM (n = 6, mean ± SD). *p < 0.05, **p < 0.01, ***p < 0.001, Student’s t-test. All the views are exactly the AGM area shown in lateral, anterior left, dorsal top. Scale bar, 50 µm. PHENOTYPE:
|
|
Cyp2aa9 regulate HSC development through the PGE2/cAMP/PKA signalling. (a-f) Inhibition of PKA by H89 resulted in reduced cmyb expression in control, but did not exacerbate the HSC phenotype in cyp2aa9 morphants. Enhancement of cAMP by Forskolin increased the cmyb expression in control, and rescued the defective HSC development in cyp2aa9 morphants. (g-j) The inhibitory effects of H89 on HSC formation could not be rescued by the injection of cyp2aa9 mRNA. (k) In vivo PGE2 concentration measured by ELISA assay. n = 3 tubes of lysates, mean ± SD, ***p < 0.001, NS, not significant, Student’s t-test. All the views of embryos are lateral, anterior left, dorsal top. |
|
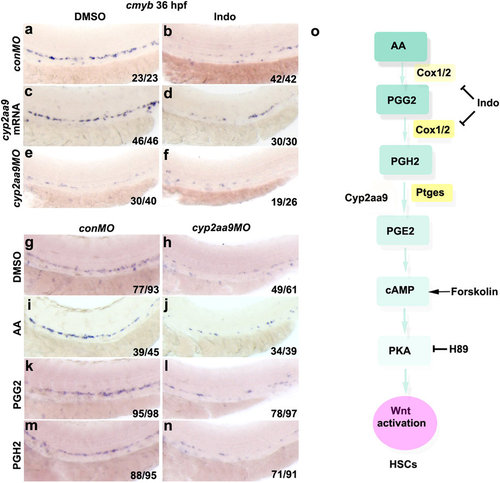
Roles of Cyp2aa9 in HSC development act at the step of PGH2-to-PGE2 conversion. (a-f) Down-regulation of cmyb expression caused by Indomethacin (Indo) could not be rescued by cyp2aa9 mRNA, and could not be exacerbated by cyp2aa9MO. (g-n) The PGE2 precursors AA, PGG2 or PGH2 could not rescue the defective HSC formation in cyp2aa9 morphants. (o) Illustration of the prostaglandin synthesis and PGE2/cAMP/PKA pathway involved in HSC development in zebrafish. Note that Cyp2aa9 acts at the step of PGH2-to-PGE2 conversion. Indo, non-selective cyclooxygenases inhibitor. Forskolin, cAMP activator. H89, PKA inhibitor. All the views of embryos are lateral, anterior left, dorsal top. |
|
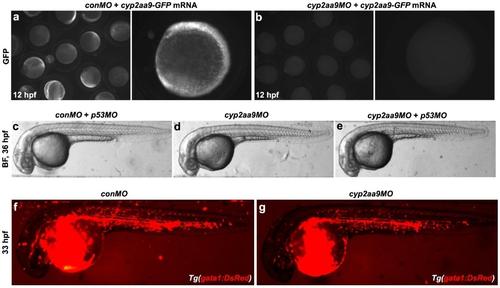
Effects of cyp2aa9MO is specific. (a-b) Translation of cyp2aa9-GFP mRNA was inhibited by cyp2aa9MO, but not conMO. (c-e) cyp2aa9MO did not lead to obvious cell apoptosis as indicated by the co-injection of p53MO, excluding off-target effects of cyp2aa9MO (c, n=30/31; d, n=24/25; e, n=26/27). (f-g) The cyp2aa9 morphants exhibited normal blood circulation under the Tg(gata1:DsRed) background (f, n=24/24; g, n=23/25). c-f, lateral views, anterior left, dorsal top. |
|
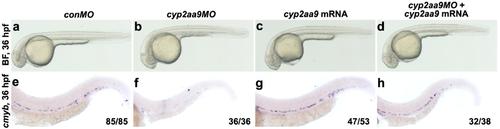
Defective cmyb expression in the cyp2aa9 morphants was rescued by the cyp2aa9 mRNA. (a-h) As indicated by the cmyb expression, defective HSC development in the cyp2aa9 morphants was rescued by the cyp2aa9 mRNA. lateral views, anterior left, dorsal top. |
|
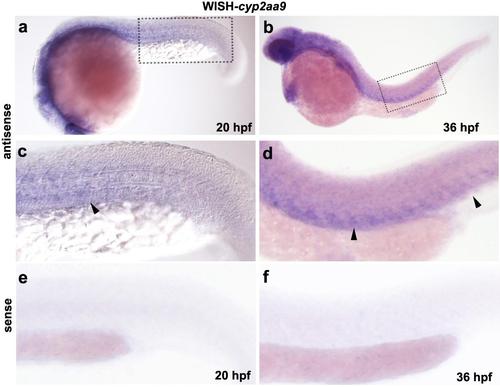
Expression pattern of cyp2aa9 in zebrafish embryos. (a-f) Whole-mount in situ hybridization indicated diffused expression of cyp2aa9 in the mesoderm and tail region (arrowheads) at 20 hpf and 36 hpf (a, n=14/17; b, n=15/17). Dashed boxs were enlarged in c-d, which is controlled by the sense probe as shown in e-f. |