- Title
-
Pax7 is required for establishment of the xanthophore lineage in zebrafish embryos
- Authors
- Nord, H., Dennhag, N., Muck, J., von Hofsten, J.
- Source
- Full text @ Mol. Biol. Cell
|
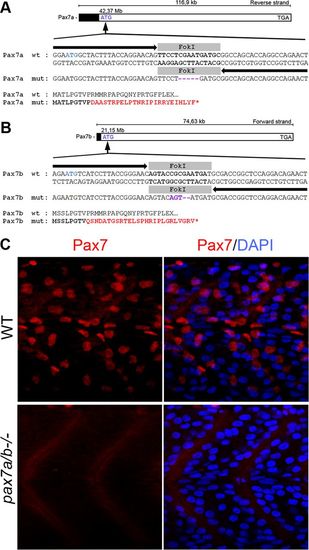
Generation of pax7a and pax7b mutants using TALENs. Schematic overview of TALEN-induced deletions resulting in frameshift mutations in genes (A) pax7a and (B) pax7b on chromosome 11. Black arrows represent the left and right TALEN sequences, and the gray box represents the spacer sequence indicating the FokI cut site. Purple indicates the insertion of mismatched nucleotides, and red indicates mismatched amino acids. (C) Lateral view of 24-hpf wt embryo and pax7a/pax7b double-mutant trunk immunofluorescently analyzed using a Pax7 antibody (red). DAPI is used to visualize nuclei. |
|
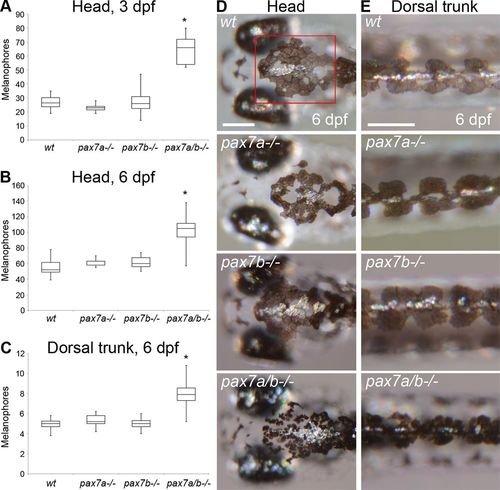
The pax7a/pax7b double-mutant embryo and larvae display an increased number of melanophores. (A) Number of melanophores on the crown of the head between the midbrain–hindbrain border and the most anterior part of the dorsal larval melanophore stripe in wt (n = 16), pax7a-/- (n = 11), pax7b-/- (n = 15), and pax7a/pax7b double-mutant (n = 11) zebrafish embryos at 3 dpf. The pax7a/pax7b double mutants differ from all other groups with p < 0.001. (B) Number of melanophores on the crown of the head between the midbrain–hindbrain border and the most anterior part of the dorsal larval melanophore stripe in wt (n = 11), pax7a-/- (n = 5), pax7b-/- (n = 7), and pax7a/pax7b double-mutant (n = 11) zebrafish larvae at 6 dpf. The pax7a/pax7b double mutants differ from all other groups with p < 0.001. The red box in D indicates area of counting. (C) Number of melanophores/somite in the dorsal larval melanophore stripe of wt (n = 12), pax7a-/- (n = 5), pax7b-/- (n = 11), and pax7a/pax7b double-mutant (n = 10) zebrafish larvae at 6 dpf. Somites 8–12 were counted. The pax7a/pax7b double mutants differ from all other groups with p < 0.001. (D) Head melanophore phenotype in wt, pax7a-/-, pax7b-/-, and pax7a/pax7b double-mutant zebrafish larvae at 6 dpf. (E) Dorsal larval melanophore stripe phenotype in wt, pax7a-/-, pax7b-/-, and pax7a/pax7b double-mutant zebrafish larvae at 6 dpf. Comparisons between groups by one-way ANOVA or Kruskal–Wallis ANOVA on ranks with Student’s–Newman–Keuls or Dunn’s post hoc contrast test for parametric and nonparametric data. Scale bar, 200 μm. PHENOTYPE:
|
|
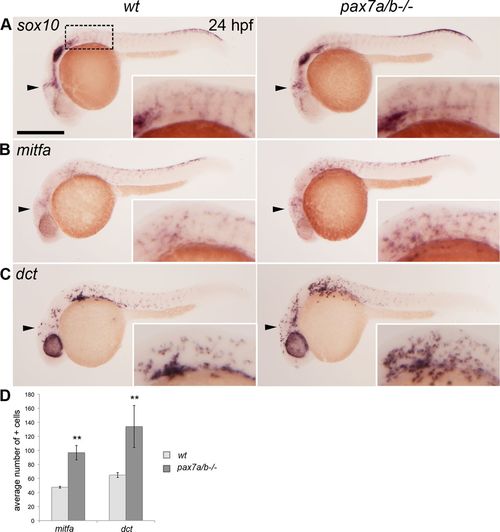
The pax7a/pax7b double mutants have an increased number of melanoblasts. Whole-mount in situ hybridization on wt siblings and pax7a/pax7b double-mutant zebrafish embryo at 24 hpf showing the expression of (A) sox10 (n > 50 for siblings and n = 7 for pax7a/pax7b double mutants), (B) mitfa (n = 5 and 5), and (C) dct (n = 7 and 3). (D) Average number of mitfa+ and dct+ cells in the region anterior to the first somite on one side of wt siblings and pax7a/pax7b double-mutant embryos at 24 hpf; positive cells in the eye were excluded. Student’s t test was used to calculate significance; **p < 0.01. Error bars indicate SEM. Box indicates area of enlargement visualized in insets. Arrowheads indicate head region where changes in expression can be detected. Scale bar, 200 μm. |
|
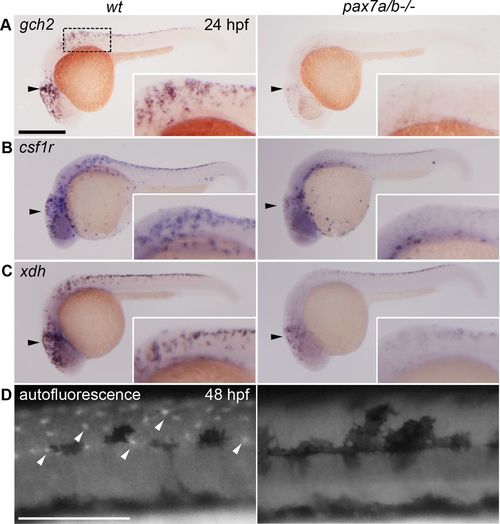
The pax7a/pax7b double-mutant embryos have a reduced xanthoblast pool and completely lack differentiated xanthophores. Whole-mount in situ hybridization on wt siblings and pax7a/pax7b double-mutant zebrafish embryo at 24 hpf showing the expression of (A) gch2 (n = 13 for siblings and n = 7 for pax7a/pax7b double mutants), (B) csf1r (n = 5 and 3), and (C) xdh (n = 3 and 5). (D) Pteridine autofluorescence in wt and pax7a/pax7b double-mutant zebrafish embryo at 48 hpf. Box indicates area of enlargement visualized in insets. Black arrowheads indicate head region where changes in expression can be detected; white arrowheads indicate a subset of autofluorescing xanthophores. Scale bar, 200 μm. |
|
Xanthophores can be rescued by specifically expressing pax7a in the neural crest cells of pax7a/pax7b double mutants. (A) Wt sibling, (B) pax7a/pax7b double mutant, and (C) pax7a/pax7b double mutant injected with a sox10:pax7a construct at the one-cell stage and reared in embryo medium with methylene blue until 5 dpf (n = 8). Boxes indicate area of magnification on the right. Arrowheads indicate a subset of methylene blue–colored xanthophores. Scale bar, 200 μm. |
|
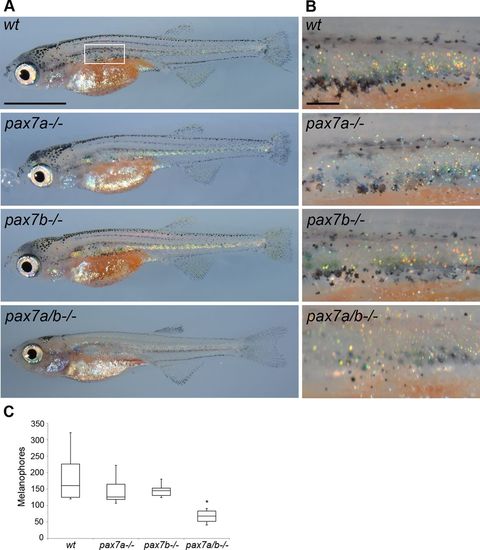
The number of melanophores is decreased in pax7a/pax7b double-mutant fish during metamorphosis. (A) Pigment phenotype of wt, pax7a-/-, pax7b-/-, and pax7a/pax7b double-mutant juvenile zebrafish at 30 dpf. Box indicates area of melanophore quantification presented in C. (B) Enlargement of area counted in C. (C) Number of melanophores in wt (n = 6), pax7a-/- (n = 8), pax7b-/- (n = 6), and pax7a/pax7b double-mutant (n = 10) juvenile zebrafish at 30 dpf. The pax7a/pax7b double-mutant zebrafish differs from all other groups with p < 0.001. Comparisons between groups by one-way ANOVA or Kruskal–Wallis ANOVA on ranks with Student’s–Newman–Keuls or Dunn’s post hoc contrast test for parametric and nonparametric data. Scale bar, 2 mm (A), 200 μm (B). PHENOTYPE:
|
|
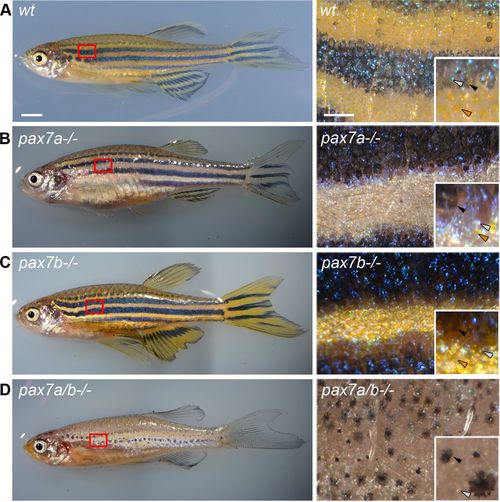
The pax7a/pax7b double-mutant adult zebrafish has few and disorganized melanophores. Pigment phenotype of 3-mo-old adult (A), wt (B), pax7a-/-, (C) pax7b-/-, and (D) pax7a/pax7b double-mutant zebrafish. Box indicates area of enlargement on the right. Insets show enlargements visualizing the presence of different types of chromatophores. Black arrowheads indicate melanophores, gray arrowheads indicate iridophores, and orange arrowheads indicate xanthophores. Scale bar, 2 mm (left), 50 μm (right). PHENOTYPE:
|