- Title
-
Liver-Enriched Gene 1, a Glycosylated Secretory Protein, Binds to FGFR and Mediates an Anti-stress Pathway to Protect Liver Development in Zebrafish
- Authors
- Hu, M., Bai, Y., Zhang, C., Liu, F., Cui, Z., Chen, J., Peng, J.
- Source
- Full text @ PLoS Genet.

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. PHENOTYPE:
|

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. PHENOTYPE:
|
|
Loss-of-function of Leg1 blocks the liver bud formation. (A) Loss of maternal zygotic Leg1a affects the liver development. Embryos were treated with or without UV25 at 24hpf, WISH was performed to assess the development of the liver (fabp10a), exocrine (trypsin) and endocrine (insulin) pancreas and intestine (fabp2a) in the maternal-zygotic leg1azju1(mu) embryos at 3.5 dpf. At least three independent WISH was performed, each time with 24–31 embryos for each sample, and representative embryos were shown. (B) WISH using prox1 and hhex to examine liver bud formation at 30 hpf in embryos treated with or without UV25 treatment at 24 hpf. Representative pictures in each group are presented. The number of embryos exhibiting the phenotype over total embryos examined are shown in the bracket. Red arrow: liver bud, blue arrow: pancreatic bud. (C and D) Images (C) and statistical analysis (D) of PH3 immunostaining to compare cell proliferation of hepatocytes in WT and maternal-zygotic leg1azju1 embryos (mu) after UV25 treatment. BHMT is an enzyme highly expressed in the liver and was used to mark out the hepatocytes. DAPI was used to stain the nuclei. Red arrows indicate PH3 positive cells in the liver. PH3 positive cells in the neural tube in WT and the mutant were also recorded in parallel. *, p<0.05. in, intestinal tube, lv, liver, nt, neural tube. EXPRESSION / LABELING:
PHENOTYPE:
|

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. |
|
N70-glycosylation is necessary for the secretion of Leg1b but not Leg1a. (A) Western blot analysis of secretion of Leg1a, Leg1aN70A, Leg1b and Leg1bN70A. Mutating N70 to A70 does not affect the secretion of Leg1aN70A but does affect Leg1bN70A. Total protein was extracted from cell pellets or supernatant. HA-tagged RNaseI was used as a control. Western blot was repeated three times.(B) Co-immunostaining of Leg1a or Leg1aN70A with the cis- and medial-Golgi marker Giantin in HepG2 cells transfected with WT leg1a and leg1aN70A plasmids, respectively. (C) Co-immunostaining of Leg1b or Leg1bN70Awith Giantin showed that Leg1b is secreted (upper panels) but Leg1bN70A is retained in the cis-Golgi network (lower panels). (D) Co-immunostaining of Leg1b or Leg1bN70A with the ER marker PDI in HepG2 cells transfected with WT leg1b and leg1bN70A plasmids, respectively. The enlarged view of the area highlighted with dashed lines are shown on the right. |
|
The compensatory mechanism is activated in leg1a mutant. (A) WISH analysis of the liver sizes in WT and maternal-zygotic leg1azju1(mu) at 3.5 and 10 dpf using the fabp10a probe. Left panel: histogram showing the percentage of normal, intermediate and small liver in the WT and leg1a mutant embryos at 3.5 and 10 dpf. The number of embryos (n) was shown on the top. Right panel: representative image of 10-dpf embryos after WISH. (B and C) leg1b expression in the zygotic mutant embryos at 4 dpf (B) and maternal-zygotic mutant embryos at 1, 3, 5 and 7dpf(C). (D) Comparison of the liver to body ratio between WT and leg1azju1mutant in the adult fish. Each dot represents the ratio for an individual adult fish. Error bar stands for the standard error. (E) Representative images of the WT and leg1azju1mutant adult fish. (F) Survival rate of the WT and leg1azju1mutant adult fish aged at 9, 14 and 18 months. The starting number of the larvae fish for each counting (SN) are shown on the top. PHENOTYPE:
|
|
Liver development in the maternal-zygotic (A) Representative images of embryos after WISH using the fabp10a probe corresponding to the result shown in Fig 1F. (B and C) Among the 32 cases shown in Fig 1G, the number of embryos exhibiting a small versus normal liver in 5 cases recorded in cold seasons (B) and 6 cases recorded in warm/hot seasons (C) were shown. (D) Plotting the liver sizes (majority normal or majority small) against daily atmospheric pressure in Hangzhou recorded during 30/12/2013 and 20/02/2015 (obtained from http://www.wunderground.com). |
|
Liver development in the maternal-zygotic leg1azju1 mutant is amenable to oxidative stress. (A) Images showing an example of determining the liver size by WISH using the fabp10a probe. WT: wild type; mu: leg1azju1 mutant; mu+UV25: leg1azju1 mutant treated with UV25. (B) WT and maternal-zygotic leg1azju1(mu) embryos were treated with 1 mJ/cm2 (UV10), 2.5 mJ/cm2(UV25) and 5 mJ/cm2 (UV50) UV at 24 hpf and grew to 3.5 dpf for WISH analysis of liver development. (C) Comparison of liver sizes between the WT and maternal-zygotic leg1azju1 (mu) embryos growing in a high density condition (200 embryos per 10-cm diameter Petri dish). (D) Growing the maternal-zygotic leg1azju1 (mu) embryos in the egg water containing 0.5% or 1% ethanol did not cause a small liver phenotype. (E) Upon UV25 treatment the maternal-zygotic leg1azju2 embryos also exhibited a small liver phenotype at 3.5 dpf. *, p<0.05, **, p<0.01, ***, p<0.001, N.S., no significance. |
|
leg1azju1 mutant does not suffer from elevated apoptosis upon UV25 treatment. Images of TUNEL assay in the 54-hpf WT and maternal-zygotic leg1azju1 embryos (mu) after UV25 treatment at 24 hpf. No abnormal apoptotic activity was observed near the endodermal region including liver (lv) and intestine (in) in the maternal-zygotic leg1azju1 embryos (mu) compared to the WT. 12 sections from six embryos for each genotype were examined. nc, notochord, nt, neural tube. |
|
Leg1a protects liver development under stress conditions through Erk signaling. (A) Injection of constitutively active form of Erk (caErk) mRNA into one-cell stage embryos impaired liver development both in WT and leg1azju1 mutant when examined with the fabp10a probe at 3.5 dpf. (B) 200 pg Cy3 labeled oligo-dT(50) was injected into the yolk at 22 hpf, and the Cy3 signal was checked at 27 hpf. CK, oligo-dT(50) uninjected control. (C) Embryos were injected with caErk or fgf8 mRNA into the yolk at 22 hpf and were then treated with UV25 at 24 hpf. The liver development in the treated embryos at 3.5 dpf was examined with the fabp10a probe at 3.5 dpf. |
|
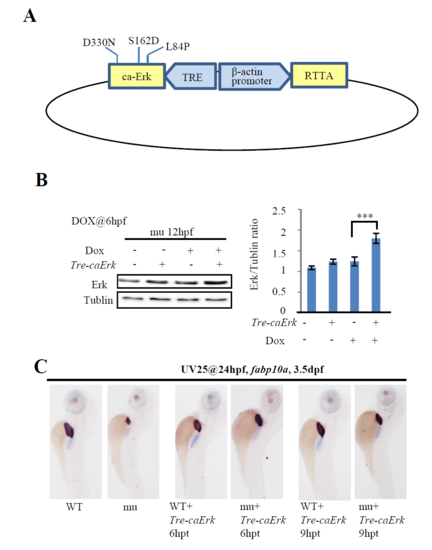
Construction of the Tre-caErk plasmid. (A) Schematic drawing showing the structure of the TRE-caErk plasmid. RTTA expression was driven by the β-actin gene promoter. Dox binds to RTTA and the Dox-RTTA complex binds to the TRE promoter to drive the expression of caErk. (B) 10 pgTre-caErk plasmid DNA was injected into one-cell stage maternal-zygotic leg1azju1 embryos (mu). These embryos were treated with Dox at 6 hpf and total protein was harvested at 12 hpf. The protein samples were subjected to western blot analysis. The total Erk versus Tubulin ratios were shown on the right. Dox, doxycycline. Error bar stands for the standard error. ***, p<0.001. Western blot was repeated three times. (C) Images of representative 3.5-dpf embryos after WISH using the fabp10a probe. Embryos was first injected with Tre-caErk plasmid at one-cell stage, then treated with UV25 at 24 hpf and followed by Dox treatment for 6 hours (6 hpt) or 9 hours (9 hpt). After Dox treatment, embryos were transferred to the normal egg water to grow to 3.5 dpf for WISH (n = 20). |
|
WISH analysis of total leg1 expression in the WT embryo at 27 hpf (A, arrow points to the endoderm region giving rise to the liver primordium) and in the WT and maternal-zygotic leg1azju1 mutant embryos at 7 dpf (B, arrow points to the liver). n=25. |
|
Immunostaining of p-Erk in the WT and maternal-zygotic leg1azju1 mutant embryos at 27 hpf. Serial cryosections (S1 to S4) from three WT embryos (WT-1, WT-2 and WT-3) and three mutant embryos (mu-1, mu-2 and mu-3) treated with UV25 at 24 hpf were shown. DAPI was used to stain the nuclei. |