- Title
-
Molecular and Cellular Characterization of a Zebrafish Optic Pathway Tumor Line Implicates Glia-Derived Progenitors in Tumorigenesis
- Authors
- Solin, S.L., Wang, Y., Mauldin, J., Schultz, L.E., Lincow, D.E., Brodskiy, P.A., Jones, C.A., Syrkin-Nikolau, J., Linn, J.M., Essner, J.J., Hostetter, J.M., Whitley, E.M., Cameron, J.D., Chou, H.H., Severin, A.J., Sakaguchi, D.S., McGrail, M.
- Source
- Full text @ PLoS One
|
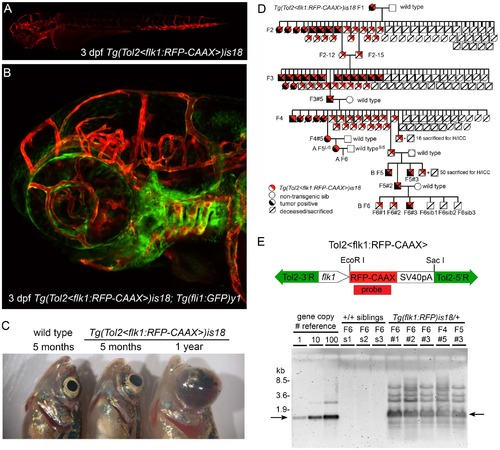
Genetic linkage of zebrafish ocular tumor model in transgenic line Tg(flk1:RFP)is18. (A) Endothelial-specific membrane targeted RFP expression in live 3 dpf zebrafish larva from transgenic line Tg(flk1:RFP)is18. (B) Endothelial-specific membrane targeted RFP expression and nuclear eGFP expression in live 3 dpf Tg(flk1:RFP)is18; Tg(fli1:eGFP)y1 zebrafish larva. (C) Gross morphology of ocular tumors in heterozygous is18/+ adults at 5 months and 1 year of age. (D) Pedigree of one family showing trans-generational linkage analysis of ocular tumor phenotype with inheritance of the Tol2<flk1:RFP> transgene in heterozygous is18/+ individuals. Two families, A and B, were established from the F4 generation. Marked individuals were used in subsequent STR/STS linkage (L/S) and Southern blot analyses (#). (E) Diagram of endothelial-specific RFP reporter construct pTol2<flk1:RFP-CAAX> with positions of EcoRI and SacI sites. Genomic Southern blot hybridized with RFP probe detects 1.3 kb EcoRI -SacI fragment (arrows) of the Tol2<flk1:RFP-CAAX> transgene. The high copy-number Tol2<flk1:RFP> transgene array is transmitted specifically to RFP expressing, tumor positive Tg(flk1:RFP)is18 progeny through the F6 generation. EXPRESSION / LABELING:
PHENOTYPE:
|
|
Histological analysis of Tg(flk1:RFP)is18 tumors reveals similarities with retinoblastoma and glioma. Coronal sections through heads of wild type (A, E–H) and Tg(flk1:RFP)is18/+ adults (B–D, I–L). (B) 5-month-old Tg(flk1:RFP)is18/+ adult with retinal tumor filling the vitreous. 1-year-old Tg(flk1:RFP)is18/+ adults with unilateral (C) or bilateral tumors (D). Tumor cells extend through the optic pathway to the tectum. (E,F) Ciliary marginal zone and mature retina in wild type. (G) Fibrillar ribbon-like structure of a wild type optic nerve exiting the eye. (H) Section through forebrain shows the sacus dorsalis and left and right lobes of the anterior region of the optic tectum. (I) Intact ciliary marginal zone in the tumorous retina from Tg(flk1:RFP)is18/+ adult shown in B. Degeneration of the retinal pigment epithelium and photoreceptor outer segments is evident. Disorganization of the retinal layers adjacent to the ciliary marginal zone is present. Streaking across the inner plexiform layer appears similar to reactive Müller glia. (J) Advanced tumors contain rosettes and blood vessels extending throughout the tumor tissue. (K) Disorganization and dysplasia in the optic nerve of the Tg(flk1:RFP)is18/+ adult in C. (L) Brain from the adult in panel D showing dysplasia of the optic tract with infiltration and disruption of normal brain structures. (M) Dysplastic areas of advanced tumor with rosettes of various sizes (arrowheads) and extensive glial fibrillar proliferation (arrow). (N) “Salt and pepper” chromatin dispersion pattern (arrowheads) consistent with a neuroectodermal tumor cell. (O) Disorganization of the optic nerve with absence of organized fibrils. (P) Expansion of the optic lobe with possible areas of necrosis (arrowheads). CMZ, ciliary marginal zone; GCL, ganglion cell layer; INL, inner nuclear layer; OL, optic lobe; ON, optic nerve; ONL, outer nuclear layer; OT, optic tract. Scale bars A, B, C, D 500 µm; E, F, G, I, J, N 20 µm; H, K, L 100 µm; M, O, P 50 µm. PHENOTYPE:
|
|
Characterization of lincRNAis18 tumors indicates a glial cell origin. Immunolabeling and in situ hybridization of cryosections from wild type retina (A–D, I, J, M–P) and advanced lincRNAis18 tumors (E–H, K, L, Q–T). Cells in rosettes in the tumors label with neurofilament marker RT-97 (red) and photoreceptor marker recoverin (green) (E, F). The synaptic vesicle marker SV2 (green), which is enriched in the retinal plexiform layers (C), was distributed throughout the fibrous tumor mass (G). (D) BrdU (green) incorporated into proliferating progenitor cells at the ciliary marginal zone of the wild type retina (arrow). (H) Intense labeling of BrdU incorporation was detected in cells forming rosettes and throughout the tumor mass. In wild type the glial marker GFAP is most evident in the Müller glia end feet that sit in the retinal ganglion cell layer (I–L). GFAP expression is absent in the oligodendrocytes and astrocytes of the optic nerve (I, K arrow). In contrast, GFAP was readily detected in long streaks throughout the tumor tissue (M, N, P), and was expressed by cells in the mutant optic nerve (O, arrow). BLBP is present at the ciliary marginal zone of wild type retina (L) and appeared present throughout the tumor mass (P). A, E, I, M, Differential interference contrast (DIC) overlay on immunofluorescence labeling images. CMZ, ciliary marginal zone; GCL, ganglion cell layer; INL, inner nuclear layer; ON, optic nerve; ONH, optic nerve head; ONL, outer nuclear layer. All scale bars represent 50 µm, except in panels K and O scale bars represent 100 µm. |
|
Examination of zebrafish lincRNAis18 expression in early development and adult tissues. (A) Nested RT-PCR showing expression of lincRNAis18 within the adult zebrafish retina. (B) In situ hybridization using non-radioactive DIG-labeled lincRNAis18 probes on adult zebrafish retina cryosections. lincRNAis18 expression is detected in the ganglion cell layer (GCL) and a subset of cells at the vitreal side of the inner nuclear layer (INL) (left, middle). Negative control, lincRNAis18 sense DIG-labeled probe (right). (C, D) RT-PCR showing relative levels of lincRNAis18 expression throughout development and in adult tissues. Control, expression of ribosomal protein S6 kinase b, polypeptide 1, rps6kb1. Blue bracket and asterisks indicate lincRNAis18 cloned and sequence confirmed products. Red bracket and asterisks indicate nonspecific products cloned and confirmed by sequencing. GCL, ganglion cell layer; INL, inner nuclear layer; ONL, outer nuclear layer; RPE, retinal pigmented epithelium. Scale bars 100 µm. |

Unillustrated author statements |