- Title
-
Hcfc1b, a zebrafish ortholog of HCFC1, regulates craniofacial development by modulating MMACHC expression
- Authors
- Quintana, A.M., Geiger, E.A., Achilly, N., Rosenblatt, D.S., Maclean, K.N., Stabler, S.P., Artinger, K.B., Appel, B., Shaikh, T.H.
- Source
- Full text @ Dev. Biol.
|
Loss of hcfc1b causes defects in craniofacial development. (A-D) Alcian:Alizarin staining was performed to visualize the developing cartilage in non-injected controls (NI), hcfc1a morphants (hcfc1a MO), hcfc1b morphants (hcfc1b MO), or embryos co-injected with hcfc1b MO and in vitro synthesized human HCFC1 mRNA. Embryos were stained at 5 days post fertilization and manual dissection of the viscerocranium and neurocranium was performed. Neurocranium is depicted in A-D and the viscerocranium is depicted in A′-D′. PHENOTYPE:
|
|
NCCs are specified and migrate correctly in morphant animals. (A and B) Dorsal view (anterior to posterior from left to right) of Tg(Sox10:mRFP) embryos injected with an hcfc1b targeting morpholino (hcfc1b-MO) were visualized for RFP expression at the 12 somite stage. (C and D) Sagittal view of non-injected (NI) or hcfc1b MO injected Tg(Sox10:mRFP) larvae at 30 h post fertilization (hpf). Arrows depict the formation of the pharyngeal arches in both wildtype and morphant animals. (E-H) In situ hybridization (ISH) of dlx2a in non-injected (NI) and hcfc1b morphants (hcfc1b MO) at 1 day post fertilization (dpf). (I and J) Lateral view of non-injected Tg(prdm1a:EGFP) (NI) or Tg(prdm1a:EGFP) embryos injected with the hcfc1b targeting morpholino (hcfc1b MO). The encircled area depicts posterior arch expression of EGFP in transgenic animals. EXPRESSION / LABELING:
|
|
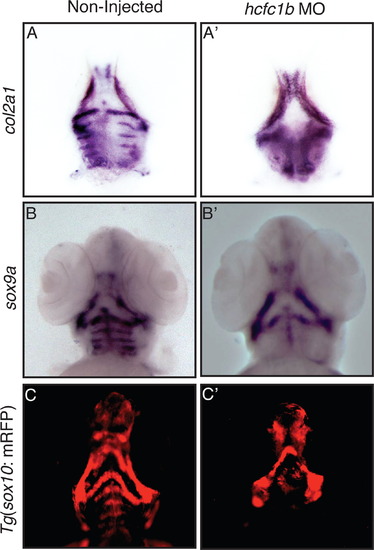
Knockdown of hcfc1b results in reduced expression of the molecular markers associated with chondrocyte differentiation. (A and B) Ventral view of RNA in situ hybridization (ISH) of col2a1 or sox9a expression in non-injected (NI) control larvae or hcfc1b morphants (hcfc1b MO) at 3 days post fertilization (dpf). (C and Có) Ventral view of non-injected (NI) control, Tg(sox10:memRFP) larvae, or transgenic larvae injected with hcfc1b morpholino (hcfc1b MO) at 3 days post fertilization. EXPRESSION / LABELING:
PHENOTYPE:
|
|
hcfc1b regulates cell proliferation. (A and B) Z-stacks for whole mount immunohistochemistry with an anti-pH3 antibody in non-injected Tg(sox10:EGFP) larvae or larvae injected with an hcfc1b targeting morpholino (hcfc1b MO). (C) Quantification of the number of pH3/Sox10 positive cells in non-injected controls or hcfc1b morphants (p=2.61431E-05, n=6 fish per group). Statistical analysis was performed using a standard T-test. EXPRESSION / LABELING:
PHENOTYPE:
|
|
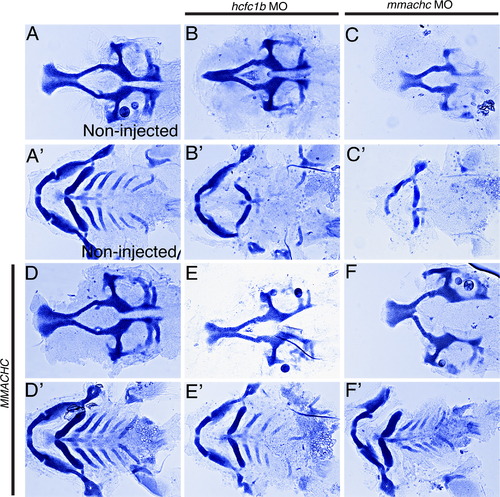
Craniofacial defects associated will loss of hcfc1b are mediated through mmachc expression. (A-F) Staining of cartilage with Alcian blue in non-injected (NI), hcfc1b morphants (hcfc1b MO), mmachc morphants (mmachc MO), human MMACHC mRNA, hcfc1b MO co-injected MMACHC mRNA, or mmachc-MO injected with MMACHC mRNA. Numbers of animals are listed in Appendix A. Embryos were stained at 4 days post fertilization and manual dissection of the viscerocranium and neurocranium was performed. Neurocranium is depicted in A-F and the viscerocranium is depicted in A′-F′. PHENOTYPE:
|
|
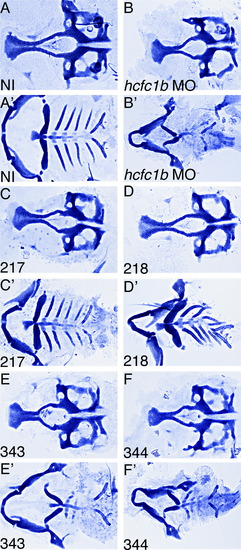
Mutations in HCFC1 differentially affect craniofacial development. (A-F) Staining of cartilage with Alcian blue on non-injected (NI), hcfc1b MO, or hcfc1b MO co-injected with the indicated mutated version of HCFC1. Numbers of animals affected are presented in Supplementary Table 5. Embryos were stained at 5 days post fertilization and manual dissection of the viscerocranium and neurocranium was performed. Neurocranium is depicted in A-F and the viscerocranium is depicted in A′-F′. PHENOTYPE:
|
Reprinted from Developmental Biology, 396(1), Quintana, A.M., Geiger, E.A., Achilly, N., Rosenblatt, D.S., Maclean, K.N., Stabler, S.P., Artinger, K.B., Appel, B., Shaikh, T.H., Hcfc1b, a zebrafish ortholog of HCFC1, regulates craniofacial development by modulating MMACHC expression, 94-106, Copyright (2014) with permission from Elsevier. Full text @ Dev. Biol.