- Title
-
Oocyte polarity requires a Bucky ball-dependent feedback amplification loop
- Authors
- Heim, A.E., Hartung, O., Rothhämel, S., Ferreira, E., Jenny, A., and Marlow, F.L.
- Source
- Full text @ Development
|
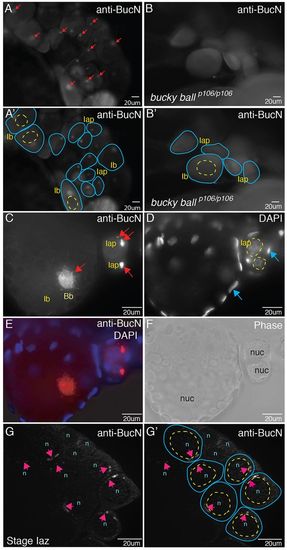
Buc protein localizes to Balbiani bodies (Bbs) and is asymmetric before Bb formation. (A,A2,C) Buc protein localization in Bbs (red arrows in A,C) of stage Iaz (zygotene), Iap (pachytene) and Ib (larger than Ia, arrested in diplotene) WT oocytes in whole-mount ovaries stained with anti-Buc antibodies. Buc protein is not detected in bucp106/p106 mutants (B,B2). (A2,B2) Tracings of the oocytes (blue lines) and their nuclei (yellow dashed lines) from A and B. (D) DAPI-labeled nuclei of oocytes (yellow dashed circles) and follicle cells (blue arrows). (E) Merge of C and D. (F) Corresponding phase image to E. (G) Perinuclear localization of Buc (pink arrows) in WT stage Iap oocytes before Bb formation. (G2) Tracing of oocytes (blue lines) and nuclei (yellow lines). n/nuc, nucleus. EXPRESSION / LABELING:
|
|
buc transgenes with introns rescue buc egg polarity phenotypes. (A,B) buc gene structure and the constructs used herein. (A) The buc promoter was used to express full-length buc or buc with a truncated 32UTR. (B) The full-length buc ORF containing the full 32UTR (cbuc) or a truncated buc 32UTR (cbuc80) without introns: grey, non-coding/intron; green, exon; light blue, 32UTR. In addition, a mutant Buc protein with the bucp106 nonsense mutation was generated (cbucp10680). (C) Schematic and genotyping assay. Products from genomic DNA are 90 bp smaller in transgenes lacking introns. Gel images of products from adult F1 progeny of cbuc80 founders. (D,E) 8- to 16-cell stage F2 progeny of a gbuc rescued mutant in different focal planes. Arrows in E indicate the excess micropyles on embryos with rescued egg polarity. (F) Clutch of gbuc rescued mutant female at 1 dpf. (G) Higher magnification of embryos from F. (H) Ventralized phenotypes of gbuc+ mutant females. (I) Rescued progeny of a gbuc80 buc mutant founder. (F-I) Rostral is left and caudal is right. (D-H) Progeny of F1 transgenic mothers. An, animal; Vg, vegetal. |
|
buc transgenes without introns disrupt egg polarity and follicle cell fates. (A-K) Dissecting microscope images. (A-E) The sibling in A shows normal egg polarity at high stage, whereas cytoplasm (black arrows) around the circumference of siblings in B indicates lack of egg polarity. (C) cbuc80 transgenic progeny resemble WT (yellow asterisks) or lack polarity (blue asterisks); high stage. (D) WT eggs (yellow arrow) have a single micropyle, whereas (E) there are multiple micropyles on the eggshells of progeny lacking egg polarity (n=1439 eggs), but not on eggs with polarity (n=2349). Homozygous WT (F) or buc/+ heterozygous (G) females lacking the cbuc80 transgene produce progeny with normal AnVg polarity, whereas (I,J) sibling WT and buc/+ females with cbuc80 produce progeny without AnVg polarity (blue asterisks). (H) buc mutants lack AnVg polarity (K) even when cbuc80 is present. (L) Quantification of phenotypes according to genotype and transgene status. Each bar represents individual F1 or F2 females. The numbers correspond to the gel in supplementary material Fig. S4. X, not in gel. (M) Quantification of egg phenotypes of cbuc progeny of F1 females. PHENOTYPE:
|
|
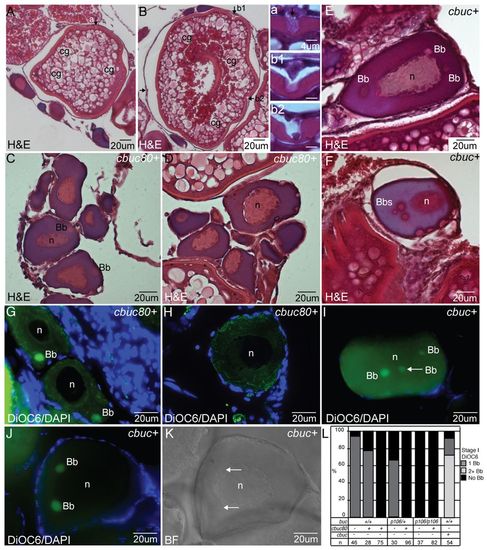
Defective Bb formation and excess polarized somatic fates in transgenics lacking introns. (A-D) Oocytes from cbuc80 transgenic founders. (A-F) Hematoxylin and Eosin (H&E)-stained F0 ovary sections reveal a normal composition of oocytes, including (A) stage III oocytes with single micropylar cells (arrow and a1), (B) stage III oocytes with multiple micropylar cells (arrows and b1 and b2), (C) stage I oocytes with Bbs and (D) stage I oocytes lacking Bbs. (E,F) Ectopic Bbs of cbuc+ F1 females. (G-J) DiOC6 staining of sectioned ovaries. cbuc80 F1 ovaries reveal primary oocytes with (G) and without (H) Bbs. (I,J) Ectopic Bbs of cbuc+ F1 females. (K) BF view of oocyte in J. Arrows indicate Bbs. (L) Quantification of Bbs from different individual F1 transgenic females labeled with DiOC6. Cg, cortical granules; n, nucleus. |
|
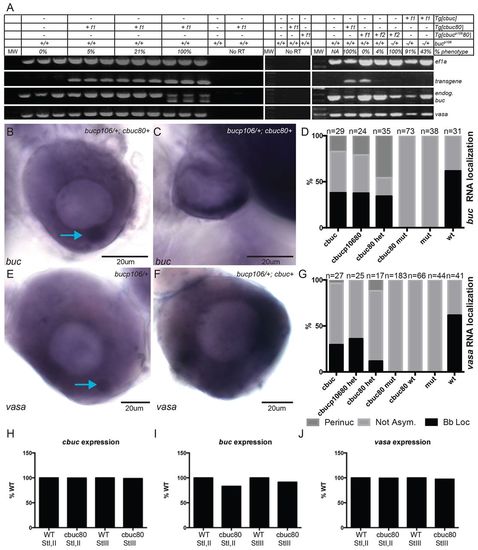
buc transgenes lacking introns disrupt RNA localization. (A) RT-PCR on ovary lysate cDNA. Transgenic and endogenous transcript expression in ovaries of F1 transgenic females expressing intron-lacking transgenes in bucp106/+ heterozygotes. (B,C) buc RNA in ovaries of cbuc80+;bucp106/+ F1 transgenic females (B) resembles WT or (C) is not asymmetrically localized. (D) Quantification of buc RNA expression patterns in stage I oocytes. (E,F) vasa expression in (E) F1 transgenic- and (F) cbuc80 F1 transgenic+ ovaries. (B,E) Blue arrows indicate the Bb. (G) Quantification of vasa expression patterns. (H-J) Relative expression of (H) cbuc80, (I) endogenous buc and (J) vasa in sorted oocytes of F1 or F2 females. (D,G) het, bucp106/+; mut, bucp106/p106; wt, buc+/+. EXPRESSION / LABELING:
|
|
Antimorphic activity of buc transgenes lacking introns requires functional protein and disrupts endogenous Buc. (A,B) Sectioned and stained oocytes. Buc protein localizes to the Bb (blue arrow) of (A) bucp106/+;cbuc80-stage Ib oocytes, and is not asymmetric in (B) bucp106/+;cbuc80+ stage Ib oocytes. (C) Quantification of Bbs of Buc-labeled oocytes of F1 or F2 females. n, the number of oocytes examined. EXPRESSION / LABELING:
|
|
bucky ball promoter drives expression in the germline and transgenic bucky ball transcripts show similar expression profiles to endogenous buc transcripts. A) Images of DNA gels show RTPCR products amplified from cDNA (+RT) produced from various tissues of cbuc80 transgene negative (cbuc80-) and positive (cbuc80+) F1 females. –RT indicates negative control for genomic contamination. Molecular weight ladder (mw), ovary (ov), head (hd), liver (liv), follicle cell (fc). buctg primers amplify transgenic transcripts, buc end primers amplify only endogenous buc, buc e+tg primers amplify both endogenous and transgenic transcripts. Aqua arrowhead indicates buc and pink asterisks indicate a nonspecific band amplified in some tissues (both were verified by sequencing). B) Images of DNA gels show RTPCR products amplified from cDNA (+RT) produced from juvenile and adult ovary of transgene negative (cbuc80-) and positive (cbuc80+) F1 females. C-E′′′) Transgenic F1 female expressing mApple under the control of the buc promoter. C-C2) Red fluorescent proteins mCherry (heart) and mApple (ovary) detected through the body wall. D-D2) In situ dissection of the Tg female revealed broad expression of mApple in early-stage oocytes throughout the ovaries. (E-E′′′) Whole-mount microscopy of dissected ovary demonstrates expression of buc promoter in early oocytes (stages I-III) (E′′-E′′′), but not in adjacent follicle cells (FC) labeled by DAPI (E2 and E′′′). |
|
bucky ball constructs comprised of exons and introns are spliced correctly. A) Schematic of constructs used to generate bucky ball transgenics with introns and exons of bucky ball. Promoters used were beta-actin and bucky ball. B) Gels showing PCR products amplified from genomic DNA using primers within the 52UTR and 32UTR of the bucky ball gene to obtain versions of buc with the introns intact and either full length or a truncated 32UTR. C) Assay for splicing of buc plasmids including exons and introns. The pBH: β-actin:gbuc construct was injected into single cell embryos along with transposase RNA. At 2dpf (a stage when endogenous buc transcripts are not detected), larvae with the integrated transgene were selected based on their fluorescent hearts and used to generate cDNA to examine the transcripts produced from the buc+introns construct. D) Schematic of spliced buc obtained from pBH: β-actin:gbuc. E) PCR fragments amplified from cDNA of pBH:β-actin:gbuc expressing embryos were as expected for properly spliced buc. Yellow asterisks indicate non-specific products amplified in the absense of buc as determined by sequencing. |
|
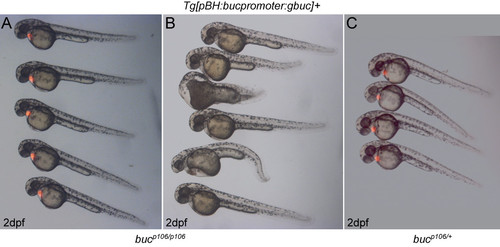
Progeny of buc mutant females are rescued by maternally supplied buc with introns. A-B) F2 embryos of a bucky ball mutant female bearing a transgene with the full buc coding sequence and introns. A) Half of the rescued progeny express the zygotic bleeding heart (red fluorescent protein in the heart) reporter; B) whereas, the other half do not express the zygotic reporter. C) Progeny of a F1 transgene positive bucky ball heterozygote expressing the bleeding heart reporter at 2 days post fertilization (dpf). |
|
Buc protein is not detected in somatic follicle cells. Images of oocytes from cbuc80 transgenic founder mothers. A-C) transgene negative or D-F) cbuc80 positive females. 40x apotome images. |
|
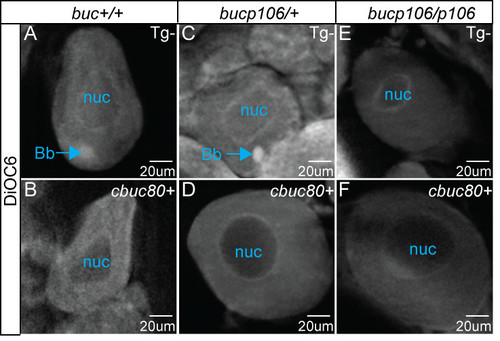
Ectopic Balbiani body formation requires the 32UTR. Images of DiOC6 labeled oocytes from cbuc80 transgenic F1 mothers. The animal (An) and vegetal (Vg) axes of eggs with normal polarity are marked accordingly. A-F) DiOC6 labels the Balbiani bodies (Bb) of A) homozygous WT and C) bucp106/+ heterozygous females. Bb formation is disrupted in both B) homozygous WT and D) bucp106/+ heterozygous genotypes when the cbuc80 transgene (Tg) is present and E, F) in bucp106/p106 homozygous mutants regardless of transgene status. Nuc denotes the nucleus. 40x apotome images. |