- Title
-
Sfrp1a and Sfrp5 function as positive regulators of Wnt and BMP signaling during early retinal development
- Authors
- Holly, V.L., Widen, S.A., Famulski, J.K., and Waskiewicz, A.J.
- Source
- Full text @ Dev. Biol.
|
BMP signaling regulates initiation of dorso-ventral retina patterning. To examine the roles of extra cellular signaling pathways in initiating dorsal retina identity, we carried out in situ hybridization for dorsal (tbx5a, bambia) and ventral (vax2) retina markers in 15 hours post fertilization (hpf) embryos. To block Wnt signaling, we utilized a heat shock inducible Wnt inhibitor Dickkopf 1b (Dkk1b). In parallel, we blocked BMP signaling by injecting embryos with morpholinos targeting growth differentiation factor 6a (gdf6a MO). Dorsal retina genes tbx5a (A-C), and bambia (G-I) are significantly reduced in the absence of BMP but not Wnt signaling. The ventral retina gene vax2 (D-F) is also unaffected by the absence of Wnt signaling, while slightly expanded in embryos lacking BMP signaling. All embryos are shown in dorsal mount with anterior aligned to the left. EXPRESSION / LABELING:
|
|
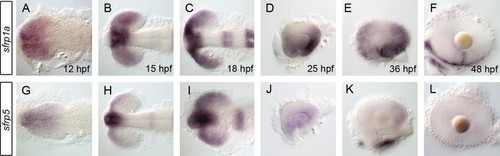
sfrp1a and sfrp5 mRNAs are expressed in the eye during retinal development. The spatio-temporal pattern of secreted frizzled related protein (sfrp) mRNA expression was assessed using whole mount in situ during zebrafish eye development. Initial expression of sfrp1a demarcates the presumptive eye field (A,B) with later stages displaying patterns specific to the presumptive ventral retina (C–E). Expression of sfrp1a persists in the ventral retina up to 48 hpf where it becomes constrained to the choroid fissure (F). In contrast, sfrp5 expression is more ventrally restricted early (G–I), then decreases significantly from 25 hpf and is absent from the retina by 48 hpf (J–L). Embryos shown are flat-mounted in dorsal (A–C,G–I) or lateral views of dissected eyes (D–F, J–L). EXPRESSION / LABELING:
|
|
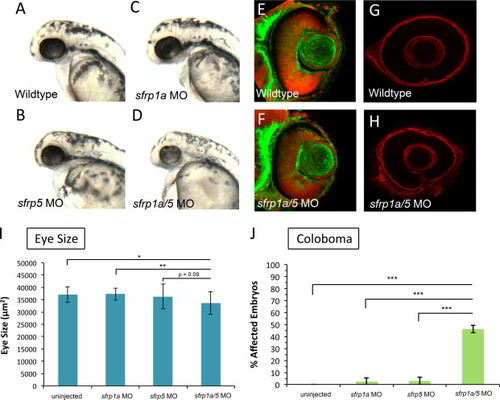
sfrp1a and sfrp5 depletion leads to small eyes and coloboma. To ascertain the function of Sfrp proteins during retinal development, we injected one-cell stage zebrafish embryos with morpholinos (MO) targeting sfrp1a and/or sfrp5. Embryos injected singly with 3 ng of sfrp1a or sfrp5 MO display overtly normal eye size at 25 hpf (A–C). In contrast, sfrp1a/5 MO co-injected embryos (3 ng each MO) display smaller eyes (D, I) (N, p<0.05; NN, p<0.01, ANOVA). 48 hpf retinas stained with phalloidin and TO-PRO3 display no overt difference between wildtype and sfrp1a/5 MO co-injected embryos (E–F). At 48 hpf WT embryos display a dissolution of Laminin staining at the choroid fissure (G). sfrp1a/5 MO embryos display persistent Laminin staining at the choroid fissure, indicating a coloboma phenotype (G,H). The increase in observed coloboma frequency is statistically significant (NNN, p<0.0036, Fisher′s Exact) when compared with wild type or embryos injected singly with Sfrp MOs (J). |
|
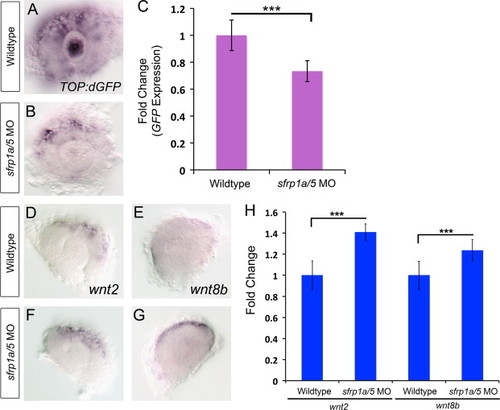
sfrp1a/sfrp5 positively regulate Wnt signaling. To determine the function of Sfrp proteins in regulating Wnt signaling, we examined wnt ligand mRNA expression and a transgene reporter of Wnt signaling using whole mount in situ hybridization and quantitative PCR. In situ hybridization for eGFP mRNA in the Tg(TOP:dGFP) line shows a decrease in eGFP expression in sfrp1a/5 MO co-injected embryos, indicating a decrease in Wnt signaling at 28 hpf (A,B). The effects of Sfrp depletion on eGFP expression were confirmed using quantitative RT-PCR (C, NNN, p<0.001). In situ hybridization of Wnt ligands wnt2 and wnt8b at 28 hpf show an expansion in the expression domain of wnt2 and wnt8b in the dorsal retina in sfrp1a/5 MO injected embryos (D–G). qRT-PCR confirmed a 1.33 fold change in wnt2 and 1.38 fold change in wnt8b expression (H, NNN, p<0.005). |
|
sfrp1a/sfrp5 regulate retinal dorso-ventral patterning. To examine dorso-ventral retina patterning in sfrp1a/5 MO co-injected embryos, we employed in situ hybridization of ventral marker genes aldh1a3, ephb3a and vax2, as well as dorsal markers aldh1a2, bambia, and tbx5a at 28 and 48 hpf. At 28 hpf, sfrp1a/5 MO injected embryos display a normal domain of ventral markers compared to WT, though with slightly reduced levels (A–F). Analysis of 28 hpf dorsal retina patterning in sfrp1a/5 MO embryos also demonstrates a decreased expression of markers (G–L). By 48 hpf, in comparison to controls, sfrp1a/5 MO injected embryos display an expansion of both aldh1a3 and vax2 expression into the dorsal regions of the retina (M–P). Analysis at 48 hpf demonstrates a clear reduction of tbx5a and bambia expression domains and levels in sfrp1a/5 MO injected embryos (Q–T). |
|
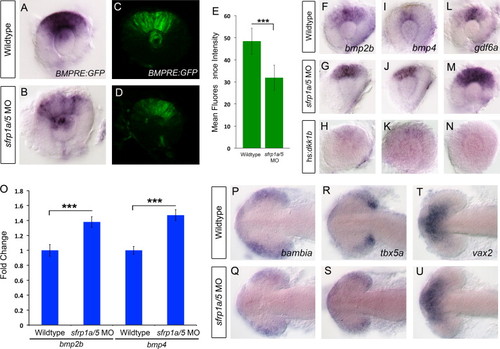
sfrp1a/sfrp5 positively regulate BMP signaling during retinal development. Given the observed alterations in dorsal retina marker gene expression, we sought to investigate the activity of BMP signaling in Sfrp depleted retinas. We measured BMP pathway activity by employing a BMP responsive transgene, Tg(BMPRE-AAV.Mlp:eGFP), which is abbreviated BMPRE:GFP. In situ hybridization for GFP mRNA, or fluorescence microscopy results indicate that compared to WT, sfrp1a/5 MO co-injected embryos display a marked decrease in GFP signal at 28 hpf (A–D). Measurements of mean retinal fluorescence intensity indicate a significant decrease in GFP signal in sfrp1a/5 MO injected embryos (E, NNN, p<0.0001). To assess expression of BMP ligand mRNAs, we conducted in situ hybridization of BMP ligands bmp2b, bmp4 and gdf6a at 28 hpf. Compared to WT, sfrp1a/5 MO injected embryos display an increase in BMP ligand expression (F–G, I–J, L–M). Conversely, inhibition of Wnt signaling by heat shock activation of dkk1b, greatly reduces expression of bmp2b, bmp4 and gdf6a at 28 hpf (H,K,N). qRT-PCR of bmp2b and bmp4 confirmed a 1.38 and 1.47 fold change in expression respectively (O, NNN, p<0.0001). To assess the functions of Sfrp proteins in the BMP-dependent initiation phase of retinal patterning, we conducted in situ hybridization for bambia and tbx5a and ventral marker vax2 during the initiation of dorsal eye patterning at 15 hpf. Compared to WT, sfrp1a/5 MO injected embryos display a decrease in tbx5a and bambia expression levels and domain while vax2 appears unaffected (P–U). EXPRESSION / LABELING:
PHENOTYPE:
|
|
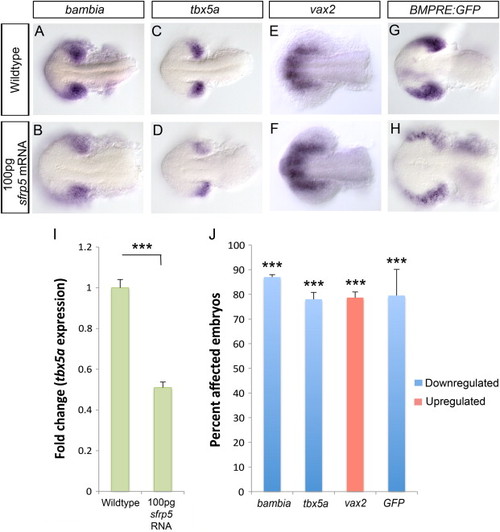
Overexpressing a high dose of sfrp5 mRNA results in loss of dorsal and increase in ventral retina identity. Using in situ hybridization, we analyzed dorsal markers bambia and tbx5a and the ventral marker vax2 at 15 hpf in embryos injected with 100 pg sfrp5 mRNA. Compared to WT, the levels of both tbx5a and bambia expression are significantly reduced in sfrp5 mRNA injected embryos (A–D) while vax2 shows both an increase in expression levels and an expansion of the domain (E–F). We measured BMP signaling using the Tg(BMPRE-AAV.Mlp:eGFP) transgenic strain, which is abbreviated BMPRE:GFP. In situ hybridization for eGFP mRNA in 15 hpf embryos injected with 100 pg sfrp5 mRNA indicates a decrease in BMP signaling compared to wildtype (G–H). Prevalence of change in expression for all probes was quantified and significance was assessed using Fisher′s Exact Test (J,NNN, p<0.0001). qRT-PCR results for tbx5a expression at 15 hpf in sfrp5 mRNA injected embryos indicate a 0.5 fold change in expression (I, NNN, p<0.0001, Student′s t-test). EXPRESSION / LABELING:
|
|
Injection of low dose (1 pg) of sfrp5 mRNA causes an increase in dorsal marker gene expression and BMP signaling. We used in situ hybridization to assess dorsal markers bambia and tbx5a and the ventral marker vax2 at 15 hpf in embryos injected with 1 pg sfrp5 mRNA. Both tbx5a and bambia expression domains are expanded in sfrp5 mRNA injected embryos compared to wildtype (A–D) while the vax2 domain is significantly reduced (E–F). We measured BMP signaling using the Tg(BMPRE-AAV.Mlp:eGFP) transgenic strain. In situ hybridization for eGFP mRNA in 15 hpf embryos injected with 1 pg sfrp5 mRNA indicate an increase in BMP signaling compared to wildtype (G–H). Prevalence of change in expression for all probes was quantified and significance was assessed using Fisher′s Exact Test (J, NNN, p<0.0001). qRT-PCR results for tbx5a expression at 15 hpf in sfrp5 mRNA injected embryos indicate a 1.17 fold change in expression (I, NN, p<0.01, Student′s t-test). EXPRESSION / LABELING:
|
|
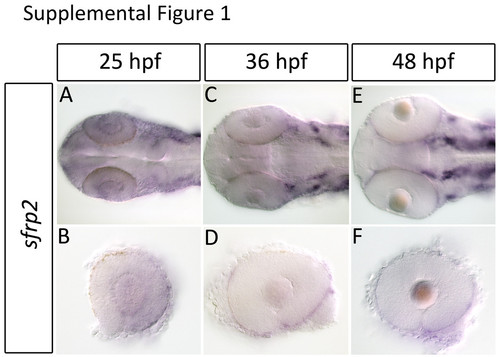
sfrp2 is expressed at low levels in the retina and primarily in the RPE during retinal development. Expression of sfrp2 during retinal development was analyzed using in situ hybridization. sfrp2 is expressed at low levels in the retina and lens at 25 hpf (A-B). By 36 hpf sfrp2 expression is confined to the RPE (C-F). |
|
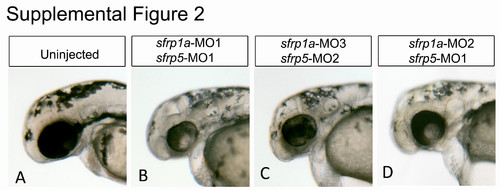
Non-overlapping sfrp1a and sfrp5 morpholinos display similar ocular phenotypes. Embryos injected with combinations of morpholino oligonucleotides (MO) (3 ng each) are analyzed for eye size and morphology at 48 hpf (A-D). The MOs were designed as either translation or splice blocking, with sfrp1a MO1 and MO2 splice blocking, and MO3 translation blocking. sfrp5 MO1 is splice blocking while MO2 is translation blocking. |
|
Fig. S3. Live embryo analysis of sfrp1a/sfrp5 depletion during retinal morphogenesis. Time lapse live confocal imaging of the Tg(Olrx3:GFP) line was employed to visualize retinal morphogenesis. Images are maximum projections of 30 slices collected every 3 μm at 10 minute intervals. Compared to WT, sfrp1a/5 morpholino injected embryos display thinning of the ventral retina as development proceeds post-24 hpf (frames 26 hpf to 39 hpf). Thinning retinal tissue is indicated with a yellow asterisk. |
|
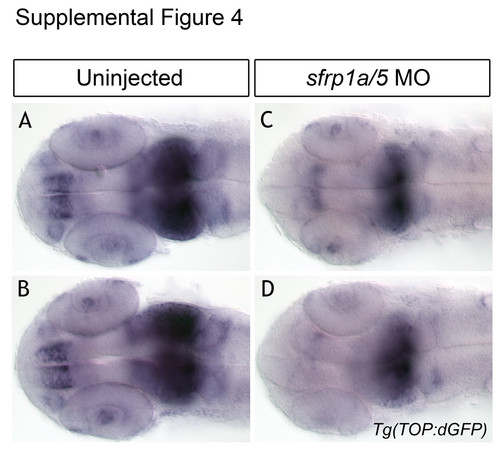
sfrp1a/5 knockdown reduces Wnt signaling in Tg(TOP:dGFP) embryos. In situ hybridization for eGFP mRNA in the Tg(TOP:dGFP) line shows a pattern of Wnt signaling regionalized in the forebrain and midbrain at 24 hpf (A-B). We note a decrease in domain and intensity of eGFP signal in sfrp1a/5 MO injected embryos (C-D), indicating a decrease in Wnt signaling in these tissues. |
|
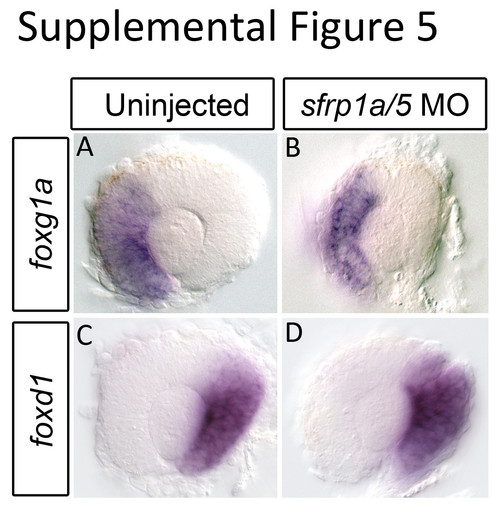
Sfrp1a/5 loss of function does not affect nasal-temporal retinal patterning. Having observed alterations in dorso-ventral patterning in sfrp1a/5 morpholino (MO) injected embryos, we sought to analyze naso-temporal pattering by performing in situ hybridization for temporal (foxd1) and nasal (foxg1a) retina markers. Compared to WT, sfrp1a/5 MO injected embryos display no change in the expression pattern of foxg1a (A-B) (n=17/17) or foxd1 (C-D) (n=14/14) at 28 hpf. EXPRESSION / LABELING:
|
|
Dorsomorphin treatment enhances BMP ligand expression. In order to analyze regulation of BMP ligand expression, BMP signaling was inhibited with 100 μM dorsomorphin. To confirm BMP inhibition tbx5a expression was analyzed using in situ hybridization. Loss of tbx5a expression at 28 hpf indicates loss of BMP signaling (A-B). Expression of BMP ligands, gdf6a and bmp4 at 28 hpf, analyzed using in situ hybridization, is increased upon dorsomorphin treatment (C-F). |
|
Morphological assessment of zebrafish embryos injected with 100 pg of sfrp5 mRNA. One-cell stage embryos were injected with a high dose (100 pg) of sfrp5 mRNA. Embryos were grown to 22 hpf and analyzed for gross morphological defects. Uninjected controls display no aberrations in either eye or alterations to axis specification (A-B, n=63/63). Of the embryos injected with a high dose of sfrp5 mRNA, 5/51 display no overt phenotype (C-D), 31/51 display subtly elongated eyes and very mild dorsalization (E-F), and 10/51 display extensive elongation of eyes as well as moderate dorsalization (G-H). |
|
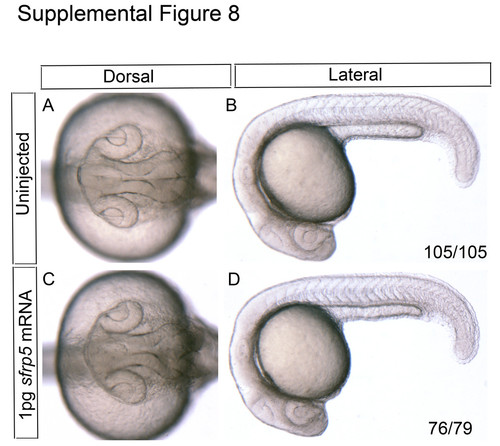
Morphological assessment of zebrafish embryos injected with 1 pg of sfrp5 mRNA. One-cell stage embryos were injected with a low dose (1 pg) of sfrp5 mRNA. Embryos were grown to 22 hpf and analyzed for gross morphological defects. Uninjected controls display no aberrations in either eye or alterations to axis specification (A-B, n=105/105). Of the embryos injected with a low dose of sfrp5 mRNA, 76/79 display no overt phenotype (C-D). |
Reprinted from Developmental Biology, 388(2), Holly, V.L., Widen, S.A., Famulski, J.K., and Waskiewicz, A.J., Sfrp1a and Sfrp5 function as positive regulators of Wnt and BMP signaling during early retinal development, 192-204, Copyright (2014) with permission from Elsevier. Full text @ Dev. Biol.