- Title
-
Six3 cooperates with Hedgehog signaling to specify ventral telencephalon by promoting early expression of Foxg1a and repressing Wnt signaling
- Authors
- Carlin, D., Sepich, D., Grover, V.K., Cooper, M.K., Solnica-Krezel, L., and Inbal, A.
- Source
- Full text @ Development
|
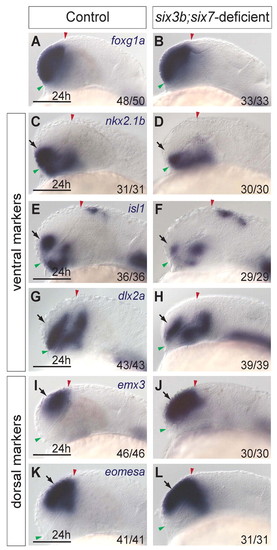
The telencephalon of six3b;six7-deficient embryos is dorsalized. (A,B) foxg1a expression in control (A) and six3b;six7-deficient (B) embryos. (C-H) Ventral telencephalic expression of nkx2.1b (C,D), isl1 (E,F) and dlx2a (G,H) in control (C,E,G) and six3b;six7-deficient (D,F,H) embryos. (I-L) Dorsal telencephalic expression of emx3 (I,J) and eomesa (K,L) in control (I,K) and six3b;six7-deficient (J,L) embryos. All embryos are at 24 hpf. Control embryos are uninjected six3bvu87/+ or six3bvu87/vu87 embryos. Arrows indicate telencephalic expression domains. Embryos are shown in lateral view with anterior towards the left. Red and green arrowheads indicate dorsal and ventral edges of the telencephalon, respectively. Fraction in each panel denotes number of embryos affected over number examined. Scale bars: 100 μm. |
|
Cellular proliferation and apoptosis do not significantly contribute to reduction of ventral telencephalon. (A-F) Expression of foxg1a (A,B), emx3 (C,D), and nkx2.1b (E,F) at the 20-somite stage in wild-type embryos treated with 2% dimethyl sulfoxide alone (A,C,E) or 20 mM hydroxyurea and 150 μM aphidicolin at 80% epiboly (B,D,F). White bracket indicates length of DV domain measured for quantification. Embryos are shown in lateral view with anterior towards the left. Red and green arrowheads indicate dorsal and ventral edges of the telencephalon, respectively. (G) Graph shows expression domain length along the DV telencephalic axis divided by average DV domain length of vehicle-treated embryos. For each sample, n=11 embryos. Blue and red columns denote vehicle- and hydroxyurea/aphidicolin-treated embryos, respectively. Error bars denote s.e.m. **P<0.01. (H,I) Expression of nkx2.1b at 24 hpf in six3b;six7-deficient embryos (H) that are also tp53zdf1/zdf1 (I). Arrows in E,F,H,I indicate ventral telencephalon. Scale bars: 100 μm. |
|
Six3b is required for specification of ventral telencephalon. (A,B) nkx2.1b expression in telencephalon (arrows) of control (A) and six3b;six7-deficient (B) embryos at the 16-somite stage. (C,D) emx3 expression in control (C) and six3b;six7-deficient (D) embryos at the 12-somite stage. (E,F) nkx2.1b (purple) expression in telencephalon (arrows) and ectopic expression (black arrowheads) at 24 hpf in Tg(hsp70l:Gal4-VP16); Tg(UAS:six3b) embryos globally misexpressing six3b (red) (F) compared with embryos not subjected to heat shock (E). GOF denotes gain of function. Embryos are shown in lateral view with anterior towards the left. Red and green arrowheads indicate dorsal and ventral edges of the telencephalon, respectively. Scale bars: 100 μm |
|
Six3b is required during early segmentation to promote ventral telencephalic fates. (A-D) nkx2.1b (purple) and six3b (red) expression in control embryos (A), six3b;six7-deficient embryos (B), six3b;six7-deficient Tg(hsp70l:Gal4-VP16); Tg(UAS:six3b) embryos induced to misexpress six3b at tailbud stage (C) and six3b;six7-deficient Tg(hsp70l:Gal4-VP16); Tg(UAS:six3b) embryos induced to misexpress six3b at the 4-somite stage (D). (E-H) isl1 (purple) and six3b (red) expression in control embryos (E), six3b;six7-deficient embryos (F), six3b;six7-deficient Tg(hsp70l:Gal4-VP16); Tg(UAS:six3b) embryos induced to misexpress six3b at tailbud stage (G) and six3b;six7-deficient Tg(hsp70l:Gal4-VP16); Tg(UAS:six3b) embryos induced to misexpress six3b at the 6-somite stage (H). Arrows indicate the ventral telencephalon in 24 hpf embryos. Embryos are shown in lateral view with anterior towards the left. Red and green arrowheads indicate dorsal and ventral edges of the telencephalon, respectively. Scale bars: 100 μm. |
|
Six3 and Hh signaling do not regulate each other during early segmentation. (A,B) Expression of ptch2 in eight-somite stage control (A) and six3b;six7-deficient (B) embryos. (C-F) Expression of six3b (C,D) and six7 (E,F) in eight-somite stage wild-type embryos treated from 6 hpf with 10 μM cyclopamine (D,F) and embryos treated with 0.1% ethanol alone (C,E). Embryos are shown in lateral view with anterior towards the left. Insets show same embryo as dorsal view with anterior towards the left. Scale bars: 100 μm. |
|
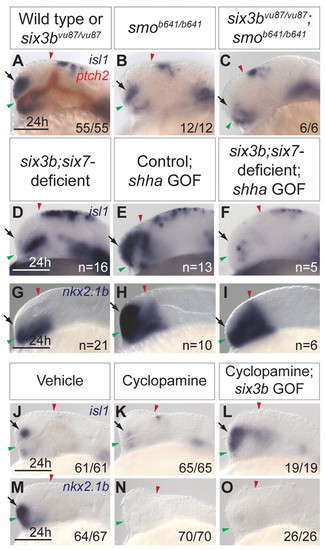
Interactions between Hh signaling and Six3 in ventral telencephalon formation. (A-C) isl1 (purple) and ptch2 (red) expression in wild-type and six3bvu87/vu87 embryos (A), smob641/b641 embryos (B) and six3bvu87/vu87;smob641/b641 embryos (C). (D-I) Expression of isl1 (D-F) and nkx2.1b (G-I) in six3b;six7-deficient embryos (D,G), control Tg(hsp70l:Gal4-VP16); Tg(UAS:shha-NH-EGFP) embryos misexpressing shha-NH-EGFP (E,H) and six3b;six7-deficient Tg(hsp70l:Gal4-VP16); Tg(UAS:shha-NH-EGFP) embryos misexpressing shha-NH-EGFP (F,I). (J-O) isl1 (J-L) and nkx2.1b (M-O) expression in vehicle-treated embryos (J,M), cyclopamine-treated embryos (K,N) and cyclopamine-treated Tg(hsp70l:Gal4-VP16); Tg(UAS:six3b) embryos misexpressing six3b (L,O). All embryos are 24 hpf. Embryos are shown in lateral view with anterior towards the left. Red and green arrowheads indicate dorsal and ventral edges of the telencephalon, respectively. Arrows indicate ventral telencephalon. Scale bars: 100 μm. |
|
Interaction between Six3 and Foxg1a in ventral telencephalon development. (A-D) Expression of foxg1a in control (A,C) and six3b;six7-deficient (B,D) embryos at the 6-somite stage (A,B) and 12-somite stage (C,D). (E-H) Expression of six3b (E,F) and six7 (G,H) in uninjected control (UIC) embryos (E,G) and MO2-foxg1a injected embryos (F,H) at the 4-somite stage. Insets in E-H are dorsal views of the same embryo with anterior leftwards. Inset scale bars: 50 μm. (I-P) isl1 (purple) (I-L) or nkx2.1b (purple) (M-P) and six3b (red) expression in UIC embryos (I,M), UIC Tg(hsp70l:Gal4-VP16); Tg(UAS:six3b) embryos misexpressing six3b (J,N), MO2-foxg1a injected embryos (K,O) and MO2-foxg1a injected Tg(hsp70l:Gal4-VP16); Tg(UAS:six3b) embryos misexpressing six3b (L,P) at 24 hpf. Embryos are shown in lateral view with anterior towards the left. Red and green arrowheads indicate dorsal and ventral edges of the telencephalon, respectively. Arrows indicate telencephalic expression domains. Scale bars: 100 μm. |
|
Six3 represses wnt8b expression in a Foxg1a-independent manner. (A-D) Expression of axin2 (A,B) and wnt8b (C,D) in control (A,C) and six3b;six7-deficient embryos (B,D) at the 8-somite stage. Black arrowhead indicates anterior limit of expression. (E-H) wnt8b (purple) and six3b (red) expression in UIC embryos (E), UIC Tg(hsp70l:Gal4-VP16); Tg(UAS:six3b) embryos misexpressing six3b (F), MO2-foxg1a-injected embryos (G) and MO2-foxg1a-injected Tg(hsp70l:Gal4-VP16); Tg(UAS:six3b) embryos misexpressing six3b (H) at 24 hpf. Embryos are shown in lateral view with anterior towards the left. Red and green arrowheads indicate dorsal and ventral edges of the telencephalon, respectively. Scale bars: 100 μm. (I) Genetic model of Six3 function in zebrafish telencephalon DV patterning. Six3 and Hh signaling function in parallel to promote foxg1a expression, which in turn promotes ventral telencephalon. Six3 and Foxg1a each can repress expression of Wnt/β-catenin ligands such as wnt8b, which can repress ventral telencephalon. |
|
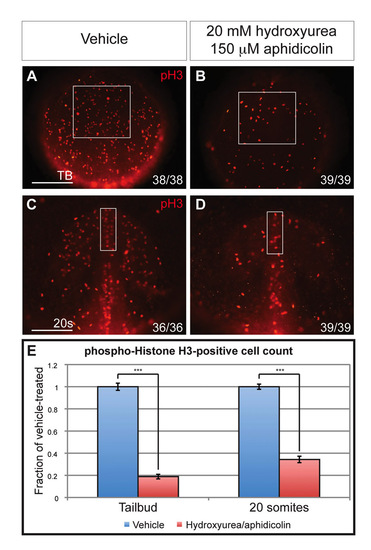
Cellular proliferation is inhibited by hydroxyurea/aphidicolin treatment. (A-D) phospho-Histone H3 labeling in tailbud stage (A,B) and 20-somite stage (C,D) embryos treated with 2% dimethyl sulfoxide alone (A,C) or with 20 mM hydroxyurea and 150 μM aphidicolin from 80% epiboly (B,D). Boxed regions mark anterior neuroectoderm (A,B) and telencephalic region (C,D). Scale bars: 100 μm. (E) Quantification of phospho-Histone H3-positive cells in the boxed regions of panels A-D divided by average cell count in vehicle-treated embryos. n=10 embryos quantified for each sample. Blue and red columns denote vehicle- and hydroxyurea/aphidicolin-treated embryos, respectively. Error bars indicate s.e.m. ***P<0.001. |
|
Apoptosis in anterior neuroectoderm is unaffected in six3b;six7-deficient embryos. (A-E) Apoptotic cells demonstrated by TUNEL in control (A,D), six3b;six7-deficient embryos (B,E) and six3b;six7;tp53-deficient embryos (C) at the eight-somite (A-C) and 12-somite (D,E) stages. Boxes outline anterior neural tube. Scale bars: 100 μm. |
|
Hh signaling promotes telencephalic isl expression in a foxg1a-dependent manner. Telencephalic isl1 expression in control embryos (A), Tghsp70l:Gal4-VP16; TgUAS:shha-NH-egfp misexpressing shha-NH-egfp (B) and MO2-foxg1a-injected control and Tghsp70l:Gal4-VP16; TgUAS:shha-NH-egfp embryos misexpressing shha-NH-egfp (C). Arrows indicate ventral telencephalon. Scale bar: 100 μm. |
|
Excess Wnt/β-catenin signaling is sufficient to repress ventral telencephalon. (A-D) isl1 (A,B) and nkx2.1b (C,D) expression in non-transgenic control embryos (A,C) and Tg(wnt8a-GFP) embryos misexpressing wnt8a-GFP after heat shock at tailbud stage (B,D). Arrows indicate ventral telencephalon. Scale bars: 100 μm. |
|
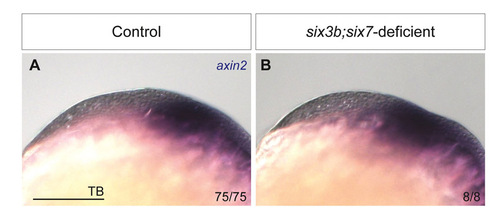
Wnt/β-catenin target axin2 is unaffected in six3b;six7-deficient embryos at tailbud stage. axin2 expression in control (A) and six3b;six7-deficient (B) embryos. 78/78 MO1-six7 injected embryos from a cross of six3bvu87/+ with six3bvu87/vu87 fish showed no observable changes in axin2 expression compared with control embryos. Scale bar: 100 μm. |