- Title
-
The osr1 and osr2 genes act in the pronephric anlage downstream of retinoic acid signaling and upstream of wnt2b to maintain pectoral fin development
- Authors
- Neto, A., Mercader, N., and Gómez-Skarmeta, J.L.
- Source
- Full text @ Development
|
Zebrafish Osr genes are necessary for pectoral fin formation. All panels show the dorsal view of 5 dpf zebrafish larvae with anterior towards the top. (A) Control uninjected embryos. (B,C) Embryos injected with 20 ng of MOosr1 (B) or MOosr2 (C). (D,E) Embryos injected with 10 ng of each morpholino. Note that the reduction of the pectoral fin is stronger in double-injected embryos (D,E) than in embryos morphant for each individual gene (B,C). Arrowheads indicate reduced fins and the asterisks indicate the complete elimination of the fins in a double morphant larvae. PHENOTYPE:
|
|
Comparative expression pattern of Osr genes and tbx5a during pectoral fin development. All panels show the dorsal view of zebrafish embryos with the anterior towards the left after whole-mount in situ hybridization using riboprobes indicated. (A-C) At 7 somites, osr1 is expressed in the IM (A, arrowhead) adjacent to the LPM domain expressing tbx5a (C, arrow). At this stage, osr2 is observed only in the posterior IM (arrowheads in A and B insets). (D-F) At 12 somites, osr1 and osr2 are found in the kidney anlage (D,E, arrowheads) medial to the fin precursor territories showing tbx5a expression (F, arrow). Asterisk in D shows a lateral displacement of osr1 expression in the posterior IM. (G-I) The same situation is observed at 24 hpf. (J-L) At 48 hpf, osr1 expression is detected very weakly in the fin bud (J), whereas osr2 is found in two patched domains within the tbx5a-expressing territory (K, arrowheads). (M-O) At 72 hpf, osr2 is expressed in a broader domain (N) overlapping with most of the tbx5a-expressing territory (O), whereas osr1 expression occurs only in small patches in the developing limbs (M, arrowheads). hpf, hours postfertilization; s, somites. EXPRESSION / LABELING:
|
|
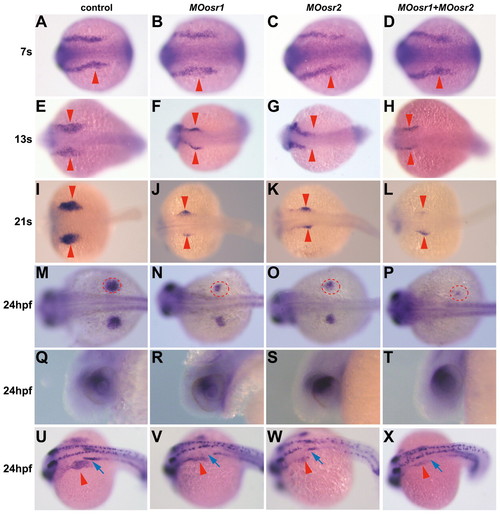
Osr genes are required for tbx5a expression during fin bud formation. Dorsal (A-P,U-X) and lateral (Q-T) views of tbx5a whole-mount in situ hybridization on zebrafish embryos injected with MOosr1, MOosr2 or a combination of both, as well as control embryos. Anterior is towards the left. (A-P) Osr MO-injected embryos reveal a smaller tbx5a expression domain in the pectoral fin field when compared with controls. Red arrowheads (A-L) or circles (M-P) mark tbx5a expression in the developing fins. (Q-T) At 24 hpf, the expression of tbx5a in the eye is not affected in morphant embryos. (U-X) tbx5a and pax2a double in situ hybridization. Red arrowheads and blue arrows indicate tbx5a and pax2a expression in the fin buds and anterior pronephros, respectively. The anterior pax2a expression domain disappears in MOosr1 and MOors1+MOosr2-injected embryos. EXPRESSION / LABELING:
|
|
Osr mRNAs rescue tbx5a and pax2a expression in double morphants embryos. Whole-mount in situ hybridization using tbx5a and pax2a riboprobes on zebrafish embryos injected with osr1/osr2 morpholinos and osr1/osr1 splicing insensitive mRNA (MTosr1, MTosr2), as indicated. Panels show a dorsolateral view at 24 hpf. In each embryo, the limb bud is encircled in red and the pronephros is indicated with an arrowhead. (A) Control uninjected embryo. (B) Embryos co-injected with MOors1 and MOosr2. (C,D) Embryos co-injected with the two Osr morpholinos and the MTosr1 (C) or the MTosr2 (D) mRNA. Co-injection of either MTosr1 or MTosr2 mRNA rescues tbx5a and pax2a expression in limb buds and pronephros, respectively. EXPRESSION / LABELING:
|
|
Osr, wnt2ba and pax2a genes are co-expressed in the kidney anlage. (A-C) Dorsal views of embryos at the 7- (A) or 12- (B,C) somite stages, showing the expression of wnt2ba (A,B) and ors2 (C), as revealed by mRNA in situ hybridization. (A) No expression of wnt2ba is visible at the 7-somite stage at the level of the intermediate mesoderm, whereas it is expressed at more anterior regions (inset). (B,C) The expression of wnt2ba is detected from the 12-somite stage (B, arrowhead) onwards in the kidney anlage, colocalizing with osr2 expression (C, arrowhead). (D-I) Transverse sections of 21-somite stage embryos with dorsal at the top. (D-F) Embryos showing the expression of osr1 (D), ors2 (E) or wnt2ba (F) genes in the kidney anlage. Inset in F shows a similar expression pattern for wnt2bb. (G-I). Embryos double stained for osr1 (G) or ors1 (H) and pax2a (Osr genes in purple and pax2a in red) and wnt2ba (purple) and osr2 (red) (I). The expression of all three genes overlaps in the pronephros (encircled). s, somite. EXPRESSION / LABELING:
|
|
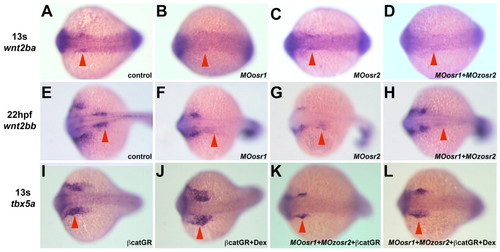
wnt2b expression in the intermediate mesoderm requires Osr function. mRNA in situ hybridizations on 13-somite stage (wnt2ba and tbx5a) and 22 hpf (wnt2bb) embryos using riboprobes as indicated on the left. All panels show dorsal views with anterior towards the left. (A-H) wnt2ba (A-D) and wnt2bb (E-H) expression in the IM (arrowheads) in wild-type (A,E), single (B,C,F,G) and double (D,H) Osr-morphant embryos. (I-L) Increasing Wnt signaling in double Osr-morphant embryos rescues tbx5a expression in the limb buds at 13 somites. (I,J) Embryos injected with β-catenin-GR mRNA show normal (I) or slightly larger (J) tbx5a expression domains (arrowheads) in the absence or presence of dexamethasone, respectively. (K,L) Embryos co-injected with β-catenin-GR mRNA and the two Osr MOs show reduced (K, arrowhead) or rescued (L, arrowhead) tbx5a expression domains in the absence or presence of dexamethasone, respectively. hpf, hours postfertilization; s, somite. EXPRESSION / LABELING:
|
|
Early tbx5a expression is reduced but not suppressed in wnt2ba morphant embryos. (A-D) Whole-mount in situ hybridization on 13-somite stage control embryos (A,B) or embryos injected with MOwnt2ba, using osr1 (A,C) or tbx5a (B,D) riboprobes. Note that whereas tbx5a expression is reduced upon wnt2ba downregulation, osr1 expression is not affected. Arrowheads highlight expression in the pronephros (A,B) or pectoral fin mesenchyme (B,D). EXPRESSION / LABELING:
|
|
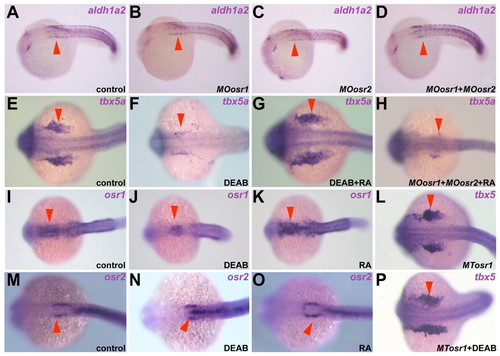
Osr gene expression is regulated by retinoic acid signaling. Dorsal views of embryos at the 21- to 24-somite stage after whole-mount in situ hybridization against the genes indicated. Anterior is towards the left. Arrowheads indicate the expression of aldh1a2 (A-D), tbx5a (E-H,L,P) and Osr (I-K,M-O) genes in the anterior somite, developing fin buds and pronephros, respectively. (A-D) aldh1a2 expression is not affected in single or double Osr-morphant embryos. (E-H) Increasing RA signaling is able to rescue tbx5a downregulation caused by DEAB (E-G, arrowheads) but not Osr impairment (H). (I-K,M-O) Reducing (J,N) or increasing (K,O) RA signaling, reduced or expanded, respectively, the expression domain of osr1 but not osr2. (L,P) Osr genes can rescue the loss of tbx5a expression when RA signaling is reduced. EXPRESSION / LABELING:
|
|
Temporal requirement of RA for tbx5a, wnt2b and Osr expression. Dorsal views of embryos at the 21- to 24-somite stage treated with DEAB from the time shown above the panels showing the expression of genes indicated on the left. (A,E,I,M,Q) Control embryos incubated with DMSO. (B,F,J,N,R) Interference with RA signaling from 50% epiboly caused strong downregulation of all genes examined. (C,G,K,O,S) Incubation with DEAB from tailbud caused strong downregulation of tbx5a, osr1, wnt2ba and wnt2bb (C,G,O,S, respectively) while not affecting osr2 expression (K). (D,H,L,P,T) Culturing embryos with DEAB from the 6-somite stage only caused some tbx5a downregulation (D). Arrowheads indicate the tbx5a-expressing domain (A-D) and the kidney anlage (E-T). EXPRESSION / LABELING:
|

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. PHENOTYPE:
|