- Title
-
Aplexone targets the HMG-CoA reductase pathway and differentially regulates arteriovenous angiogenesis
- Authors
- Choi, J., Mouillesseaux, K., Wang, Z., Fiji, H.D., Kinderman, S.S., Otto, G.W., Geisler, R., Kwon, O., and Chen, J.N.
- Source
- Full text @ Development
|
The development of the zebrafish caudal vein plexus. (A-I) Confocal images of caudal vasculature at different developmental stages. (A-E) Untreated control Tg(kdrl:GFP) embryos. (F-I) Tg(kdrl:GFP) embryos treated with 10 μM aplexone beginning at 10 hpf. The red arrow in A indicates a primary ISV. The yellow arrowhead in B indicates venous angiogenic sprouts. Yellow arrows in G,H indicate filopodia that fail to mature into angiogenic sprouts. Insets in B,G,H are magnified images of the areas indicated by the yellow arrowhead and arrows. Red bars in B and E indicate the distance between the axial artery and vein. (J,K) Optical cross-sections of blood vessels in control (J) and aplexone-treated (K) embryos at 48 hpf. Yellow bars in D and I indicate the position where the cross-sections were extracted. (J′,K′) Schematic drawings of the cross-sections in control (J′) and aplexone-treated (K′) embryos. Red represents the dorsal aorta and blue the caudal vein. (L,M) Confocal images of blood vessels in the trunk of control (L) and aplexone-treated (M) Tg(kdrl:GFP) embryos. Images were taken at 48 hpf. (L′,M′) Schematic drawings of L and M. Red and blue represent the artery and vein, respectively. Horizontal vessels are the dorsal aorta and posterior cardinal vein. Vertical vessels are intersegmental vessels. |
|
Aplexone does not affect arterial and venous fate. Untreated control embryos (A,A′,C,C′,E,E′) and embryos treated with 10 μM aplexone beginning at 10 hpf (B,B′,D,D′,F,F′) were fixed at 24 hpf. (A-F′) Arterial and venous fates were analyzed by in situ hybridization using ephrinB2a (A,A′,B,B′), ephB4 (C,C′,D,D′) and flt4 (E,E′,F,F′) probes. Arrowheads indicate the dorsal aorta. Embryos were analyzed at higher magnification in the trunk in A′-F′, with regions of interest indicated by black boxes in A-F. Arrows indicate the posterior cardinal vein. |
|
Aplexone affects the migration of caudal vein endothelial cells. (A,B) Representative images of the caudal vein of control (A) and 10 μM aplexone-treated (B) embryos at 30 hpf. Images were overlaid with the endothelial cell migration path inferred from time-lapse confocal movies taken from 25 hpf to 30 hpf. The migration of caudal vein endothelial cells was traced using the Manual Tracking feature of ImageJ and the positions of ISV sprouting points were used as references to adjust for the growth of embryos. (C) The distance of endothelial cell migration in caudal vein of control and aplexone-treated Tg(kdrl:GFP) embryos. Asterisk indicates P<0.05. (D) The window of application of aplexone. Gray bars represent the developmental stages during which Tg(kdrl:GFP) embryos were exposed to 10 μM aplexone and the caudal vein phenotype was analyzed at 48 hpf. + indicates that caudal vein angiogenesis was inhibited. – indicates the formation of normal caudal vein plexus. |
|
Blood circulation is not required for sprouting angiogenesis from the caudal vein. (A,B) Caudal vein plexus in wild-type Tg(kdrl:GFP) embryos observed at 32 hpf (A) and 48 hpf (B). (C-F) At 32 hpf, the primordial caudal vein plexus was properly formed in tnnt2 morphant (C) and smo mutant (E) embryos, but the plexus structure was not maintained and degenerated into a single-lumen structure at 48 hpf (D,F). (A′-F′) Optical cross-sections of blood vessels indicated by yellow lines in A-F. (A"-F") Schematic drawings of images shown in A′-F′. Red and blue represent the artery and vein, respectively. |
|
Aplexone affects the HMGCR pathway. (A) Simplified schematic diagram of the HMGCR pathway. Enzymes involved in the pathway are shown in blue. Feedback loop is indicated in gray. Chemical inhibitors of the pathway are indicated in red. HMGCS, 3-hydroxy-3-methylglutaryl-CoA synthase; HMGCR, HMG CoA reductase; FDPS, farnesyl diphosphate synthetase; Cyp51, lanosterol 14 α-demethylase; SC4MOL, sterol-C4-methyl oxidase-like; DHCR7, 7-dehydrocholesterol reductase; INSIG, insulin induced gene 1; SREBP, sterol regulatory element binding proteins. (B) Relative cholesterol levels in embryos treated with 0.4% DMSO as a control (DMSO), aplexone (AP, structure shown in C), atorvastatin (AS) or a non-functional aplexone analog (AP13, structure shown in D). Both aplexone and atorvastatin reduce cholesterol levels significantly (*P<0.05). Data are mean±s.e.m. (E-J) Aplexone and atorvastatin have a synergistic effect on caudal vein angiogenesis. Embryos were treated with the chemicals indicated beginning at 10 hpf and caudal vein images were taken at 48 hpf. (K-M) Mevalonate reverses the effect of aplexone on caudal vein plexus formation. Caudal vein plexus of embryos treated with 5 μM aplexone beginning at 2 hpf (K), injected with 1 nl of 2 M mevalonate (L) or injected with mevalonate followed by aplexone treatment (M). |
|
Aplexone inhibits caudal vein angiogenesis by blocking geranylgeranylation. (A) Caudal vein of control Tg(kdrl:GFP) embryo at 48 hpf. (B-D) Caudal vein of L744,832-injected (B), GGTI-2133-injected (C) or 50 μM rockout-treated (D) Tg(kdrl:GFP) embryos analyzed at 48 hpf. (E,F) Representative confocal images of the localization of mCherry-rhoCAAX fusion proteins in control (E) and aplexone-treated (F) embryos at 80% epiboly. mCherry-rhoCAAX fusion proteins are predominantly localized to the plasma membrane in control embryos but are mislocalized to the nucleus and cytoplasm in aplexone-treated embryos. (G) Table quantifying the subcellular localization of mCherry-rhoCAAX in control or aplexone-treated embryos. |
|
Aplexone inhibits migration of germ cells, cardiomyocytes and arterial endothelial cells. (A,B,C) Overall phenotype of control and aplexone-treated embryos at 30 hpf. (A2,B′,C′) Vasculature in trunk. Aplexone (10 μM) does not affect the development of primary ISVs but 30 μM aplexone inhibits the growth of primary ISVs. (D,E) Germ cells in control (D) and aplexone-treated (E) embryos at 24 hpf were detected by in situ hybridization using the nanos probe. (F) The percentage of embryos with ectopic primordial germ cells in control (n=26), 5 μM (n=13) and 10 μM (n=14) aplexone-treated embryos. (G,H) Cardiomyocytes migrate to the midline and form a heart tube in Tg(myl7:GFP) embryos by 24 hpf (G), but fail to migrate to midline in embryos treated with 30 μM aplexone (H). (I) The percentage of embryos with cardia bifida in control (n=16) and 10 μM (n=17), 20 μM (n=17) and 30 μM (n=18) aplexone-treated embryos. |
|
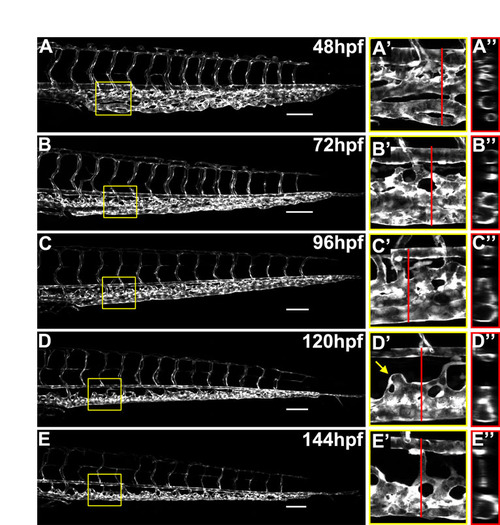
Remodeling of the caudal vein plexus. (A-E) Confocal images of caudal vasculature from 2 dpf to 6 dpf. (A′-E′) Higher magnification images of the boxed regions shown in A-E. (A′′-E′′) Optical cross-sections of blood vessels indicated by red bars in A′-E′. |