- Title
-
Sex Reversal in Zebrafish fancl Mutants Is Caused by Tp53-Mediated Germ Cell Apoptosis
- Authors
- Rodriguez-Mari, A., Cañestro, C., Bremiller, R.A., Nguyen-Johnson, A., Asakawa, K., Kawakami, K., and Postlethwait, J.H.
- Source
- Full text @ PLoS Genet.
|
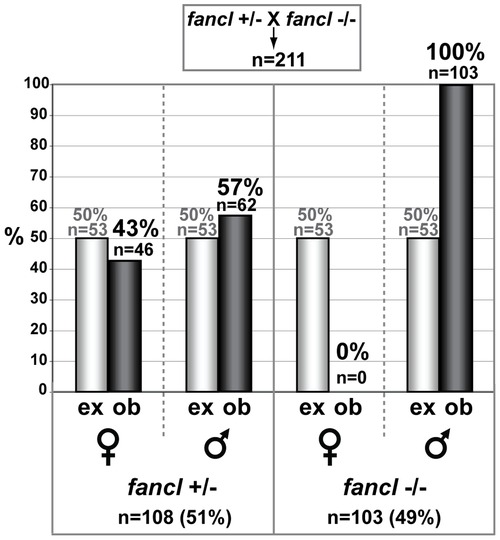
The absence of females in fancl homozygous mutants is due to sex reversal. The bar graph represents percentages of expected (ex, grey bars) and observed (ob, black bars) females and males among 211 progeny from a cross of fancl heterozygous females (fancl+/-) to fancl homozygous mutant males (fancl-/-). Total numbers (n) and percentages (%) of animals in each category are indicated on the graph. The expected ratio of female heterozygotes to male heterozygotes to female homozygous mutants to male homozygous mutants is 1:1:1:1, but we observed a ratio of about 1:1:0:2 (46 fancl+/- females: 62 fancl+/- males: 0 fancl-/- females: 103 fancl-/- males). This result rules out the hypothesis that homozygous mutant females die, but is predicted by the hypothesis that homozygous mutants that otherwise would have become females develop instead as males. PHENOTYPE:
|
|
Zebrafish germ cells express fancl during gonad development. In situ hybridizations with fancl probe were performed on cryo-sections of wild-type animals at different stages of gonad development. Weak fancl expression signal (arrows) was detected in undifferentiated gonads at 17 days post-fertilization (dpf) (A) and 23 dpf (B). Signal became stronger in germ cells (arrows) of transitioning and immature ovaries (ooplasm of oocytes, arrows in C,E,G) and transitioning and immature testes (D,F,H) at 26, 33, and 37 dpf. In adults, fancl expression was restricted to germ cells, but signal intensity depended on the stage of germ cell differentiation. In adult ovaries (I), early stage IB oocytes (eIB) already showed low fancl expression and late stage IB oocytes (lIB) showed strong fancl signal in the ooplasm, suggesting that fancl expression initiated in early stage IB oocytes. As oogenesis progressed, ooplasm volume increased, cortical alveoli appeared (stage II), yolk accumulated (stage III), and fancl expression signal became diluted. In adult testes (J), fancl expression signal was detected in a subset of cells with large nuclei and morphology consistent with primary spermatocytes (sc), but signal was not detected in cells with small nuclei in an advanced stage of spermatogenesis (i.e. spermatids and sperm (sp)). Oocyte staging is according to [49] and [29]. Scale bar: 0.1 mm. EXPRESSION / LABELING:
|
|
Gonads of fancl mutants have germ cells but fail to maintain a female gene expression profile. Comparative expression analysis of the germ cell marker vasa, the early female somatic cell marker cyp19a1a, and the early male somatic cell marker amh in developing gonads of fancl homozygous mutants (fancl) and their wild-type sibling controls (WT), and in wild-type animals depleted of germ cells by dead end morpholino knockdown (dnd). To monitor the expression of vasa, cyp19a1a and amh, in situ hybridization (ISH) experiments were performed on adjacent cryo-sections of each animal at different stages of gonad development: undifferentiated gonads at 19 dpf (A–I), transitional juvenile gonads at 26 dpf (J–X) and post-transitional juvenile gonads at 33 dpf (Y-M′). Arrows point to examples of regions showing gene expression. ISH with vasa probe labeled germ cells in wild types (A,J,M,Y,B′) and fancl mutants (D,P,S,E′,H′), and confirmed the depletion of germ cells in dnd animals (G,V,K′). In undifferentiated gonads at 19 dpf, female and male markers were expressed in all genotypes: WT (B,C), fancl (E,F) and dnd knockdown animals (H,I), showing that the onset of cyp19a1a and amh expression does not depend on germ cells or on fancl function. At 26 dpf, controls had started to enter either the male pathway by down-regulating cyp19a1a and up-regulating amh (K,L) or conversely into the female pathway by up-regulating cyp19a1a and down-regulating amh (N,O), correlated with the presence of few or many oocytes, respectively. In contrast, most 26 dpf fancl mutants already showed a male expression profile by the absence of cyp19a1a and the up-regulation of amh (Q,R) and only one fancl mutant showed a low number of cyp19a1a-expressing cells while nevertheless maintaining high amh expression (T,U). Except for vasa, expression profiles of 26 dpf dnd knockdown gonads were similar to fancl mutants (W,X). At 33 dpf, wild-type controls showed either a male expression profile (no cyp19a1a and high amh expression, Z,A′) or a female expression profile (high cyp19a1a and no amh expression, C′,D′). Most 33 dpf fancl mutants showed a male expression profile (F′,G′), even if gonads maintained an ovary-like morphology (I′,J′). All 33 dpf dnd animals showed a male expression profile (L′,M′). Scale bar: 0.1 mm (A). EXPRESSION / LABELING:
|
|
Juvenile gonads of fancl mutants contain oocytes that fail to progress through meiosis. Histological comparison of germ cell development in fancl homozygous mutants (fancl) and wild-type sibling controls (WT), by hematoxylin and eosin staining of gonads at different stages of development: undifferentiated, 19–22 dpf (A,B), transitional juveniles, 26 dpf (C–F), post-transitional juveniles, 32 dpf (G–I) and adults (J–L). At 19–22 dpf, no morphological differences were observed between WT (n = 10) and fancl animals (n = 10) and both genotypes showed early stage IB perinucleolar oocytes (epo in A,B). At 26 dpf, the first histological differences between WT and fancl became apparent. Most WT controls (7 out of 9 individuals) had abundant enlarged perinucleolar oocytes that had progressed from early stage IB to late stage IB (lpo in C), and only two lacked late stage IB oocytes (E). In contrast, only one of twelve fancl mutant animals had enlarged oocytes at late stage IB (lpo in D), while the majority (11 out of 12) lacked late stage IB oocytes (F). Both wild-type and fancl mutant gonads that lacked oocytes possessed spermatogonia (sg in E,F). Remarkably, in contrast to wild types, fancl mutants showed abundant pyknotic cells (pc) at 26 dpf (D,F). At 32 dpf, gonads showed unmistakably the morphology of either ovary or testis, and in wild-type controls, approximately half of the animals (6 out of 11) had ovaries with perinucleolar oocytes at late stage IB (lpo in G) and the other half (5 out of 11) showed the typical testis morphology with abundant spermatogonia (sg) and spermatocytes (sc) arranged in cysts (dashed line in H). In contrast to controls, all fancl mutants (n = 8) had gonads that lacked perinucleolar oocytes, and showed testis morphology with groups of spermatogonia (sg) and spermatocytes (sc) (I). Finally, in adults, half of the WT controls (5 out of 10) had mature ovaries filled with oocytes at different stages of oogenesis: stage IA, IB, II, III and IV (J), and the other half (5 out of 10) had mature testes (K), in contrast to fancl mutants in which all animals (n = 7), had mature testes filled with germ cells at different stages of spermatogenesis: spermatogonia (sg), spermatocytes (sc) and sperm (sp) (L), and none of the seven fancl mutants had ovaries. Oocyte stages described according to [49]; Spermatogenesis stages described according to [28]. Scale bar: 0.02 mm (as in A, except for J). PHENOTYPE:
|
|
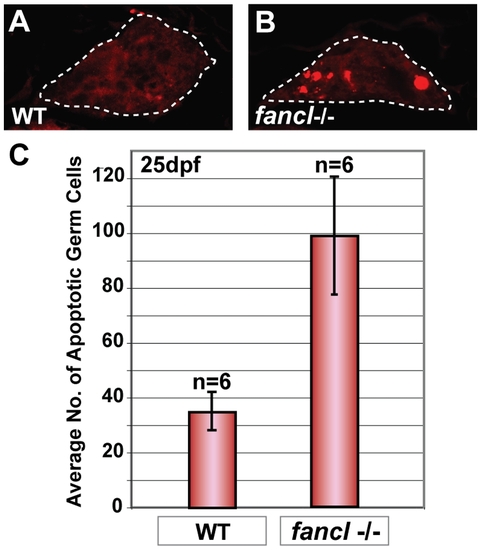
Increased germ cell apoptosis in fancl mutants at 25 dpf. Immunodetection of apoptosis by anti-active Caspase-3 in paraffin sections of gonads of wild-type sibling controls (WT) and fancl homozygous mutants (fancl-/-) at 25 dpf (A,B). Presence of Caspase-3-positive cells (shown in red) was lower in gonads of WT (A) than in fancl mutants (B). Gonads outlined by a dashed line (A,B). Bar graph representing the average number of Caspase-3-positive germ cells in each genotype: wild-type sibling controls (WT; n = 6) and fancl homozygous mutants (fancl-/-; n = 6) at 25 dpf (C). Results showed that the average number of apoptotic germ cells in fancl mutants (x- = 99±43) was about three fold higher than in wild-type sibling controls (x- = 35±14), revealing an abnormal increase of germ cell apoptosis in fancl mutants at 25 dpf, a critical period for sex determination (C). PHENOTYPE:
|
|
Mutation of tp53 rescues the female-to-male sex-reversal phenotype of fancl mutants by reducing germ cell apoptosis. (A) The distribution of individuals of different tp53 genotypes among fancl-/- homozygous mutant progeny (n = 44) from an in-cross of double heterozygotes (fancl+/-;tp53+/-) is shown in a bar graph representing the number of females (purple bar) and males (green bars) distributed according to their tp53 genotypes (wild type, heterozygous or homozygous mutant). Rescue of female-to-male sex reversal was observed exclusively in fancl-/- mutant homozygotes that were also homozygous for the tp53 mutation (n = 15): 11 fancl-/-;tp53-/- animals developed as females and 4 developed as males. No rescue was observed in fancl mutants that were either wild-type (n = 8; fancl-/-;tp53+/+) or heterozygous for the tp53 mutation (n = 21; fancl-/-;tp53+/-), which all developed as males. Total numbers of animals (n) are indicated on the graph per each sex in each genotype. (B,C) Hematoxylin and eosin staining of gonad sections of wild-type female (fancl+/+;tp53+/+, B) and rescued female doubly homozygous mutant (fancl-/-;tp53-/-, C) at adult stage, revealed the presence of morphologically normal ovaries in the rescued fancl-/-;tp53-/- females. Ovaries of both genotypes had oocytes at different stages of development (i.e.: IB, II, III, IV). Scale bar: 0.1 mm (B,C). (D,E,F) tp53 mutation reduces germ cell apoptosis in fancl mutants at 25 dpf. Immunodetection of apoptosis by anti-active Caspase-3 in paraffin sections of gonads of fancl homozygous mutants simultaneously homozygous wild-type (D) or homozygous mutant for tp53 (E) at 25 dpf. Dashed lines outline gonad boundaries (D,E). (F) Bar graph representing the average number of Caspase-3-positive germ cells in fancl-/-;tp53+/+ (n = 5) and fancl-/-;tp53-/- (n = 5) at 25 dpf. Results showed that the average number of apoptotic germ cells was approximately three fold lower in doubly homozygous mutant animals (fancl-/-;tp53-/-; x- = 30±56) than their fancl-/- mutant siblings that were wild-type for tp53 (fancl-/-;tp53+/+; x- = 105±71). This result shows that tp53 mutation decreased the number of apoptotic germ cells in fancl mutants at 25 dpf and demonstrates that the abnormal increase in germ cell apoptosis in fancl mutants that compromised the survival of developing oocytes was the mechanism responsible for the female-to-male sex reversal. PHENOTYPE:
|

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. PHENOTYPE:
|