- Title
-
Selective interaction between Trf3 and Taf3 required for early development and hematopoiesis
- Authors
- Hart, D.O., Santra, M.K., Raha, T., and Green, M.R.
- Source
- Full text @ Dev. Dyn.
|
Taf3 is required for normal development and hematopoiesis in early zebrafish embryos. A: RT-PCR analysis monitoring expression of taf3 and, as a control, gapdh in zebrafish embryos at 0, 4, 8, and 24 hpf. B: Immunoblot analysis monitoring expression of Taf3 in zebrafish embryos at 0, 4, 6, 18, and 24 hpf. C: Whole-mount in situ hybridization with a riboprobe to taf3 at 6 and 24 hpf. D: Phenotypic analysis of embryos injected with a control, taf3, trf3, or cdx4 MO at 24 hpf. E: (Top) Whole-mount in situ hybridization with riboprobes to gata1 and pax2 (14 hpf). (Bottom) Fluorescence microscopy monitoring expression of a gata1:dsRed transgene at 30 hpf. F: Whole-mount in situ hybridization with riboprobes to scl and hbbe1 (18 hpf). EXPRESSION / LABELING:
PHENOTYPE:
|
|
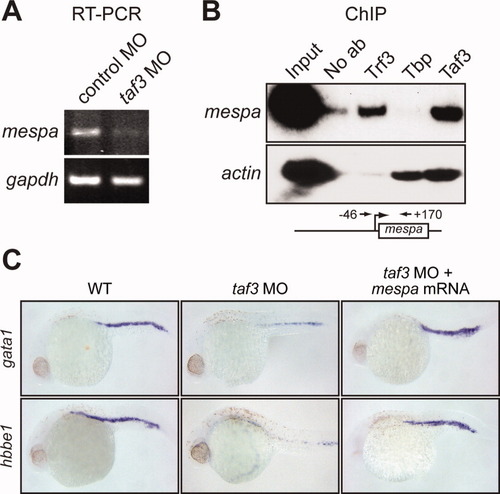
Taf3 is selectively recruited to the promoter of the Trf3 target gene mespa in vivo and is required for mespa expression. A: RT-PCR analysis monitoring expression of mespa in control and taf3 MO-treated embryos at 6 hpf. B: ChIP analysis monitoring occupancy of Trf3, Tbp, and Taf3 at the mespa and actin promoters in embryos at 6 hpf. Control ChIP experiments were performed in the absence of antibody (No ab). A schematic representation of the mespa promoter showing the location of the primer pairs used for ChIP analysis is shown (bottom). C: Whole-mount in situ hybridization with riboprobes to gata1 and hbbe1 (28 hpf) in embryos injected with a control MO or taf3 MO, or in taf3 MO-treated embryos injected with a mespa mRNA. |
|
Taf3 selectively interacts with the C-terminal domain of Trf3 but not Tbp. A: Co-immunoprecipitation assays. Plasmids expressing Flag-tagged Taf3 and either myc-tagged Trf3, Tbp, or Trf3/Tbp chimeric protein were co-transfected into COS-7 cells. Taf3 was immunoprecipitated with an anti-Flag antibody, and the immunoprecipitates were analyzed for the presence of Trf3, Tbp, or Trf3/Tbp chimera by immunoblotting with an anti-myc antibody. B: Co-immunoprecipitation assays as described in A using HA-tagged TAF1. C: Schematic diagram of the zebrafish Trf3 protein, showing the substitution mutations. D: Co-immunoprecipitation assays. Plasmids expressing Flag-tagged Taf3 and either myc-tagged Tbp, wild-type Trf3, or Trf3 mutant were co-transfected into COS-7 cells. Tbp or Trf3 was immunoprecipitated with an anti-myc antibody, and the immunoprecipitates were analyzed for the presence of Taf3 by immunoblotting with an anti-Flag antibody. |
|
Interaction with Taf3 is required for Trf3 function in zebrafish development. A: Whole-mount in situ hybridization with a riboprobe to hbae1 (22 hpf) in trf3 MO-treated embryos injected with mRNAs expressing tbp, trf3, or a trf3 mutant. Arrowheads denote staining in the intermediate cell mass (ICM). B: Whole-mount in situ hybridization with riboprobes to gata1 or pax2 (14 hpf). C: RT-PCR analysis of scl1, lmo2, or, as a control, gapdh expression at 18 hpf. EXPRESSION / LABELING:
PHENOTYPE:
|
|
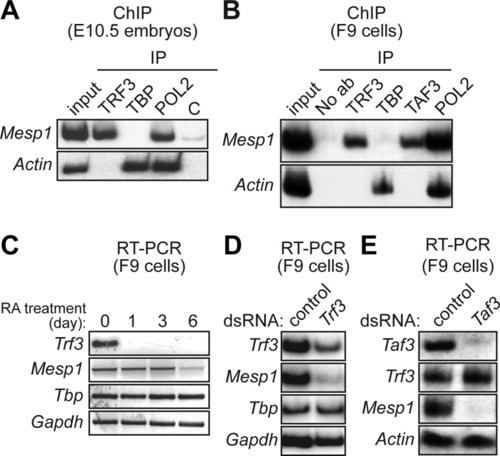
Requirement of TRF3 and TAF3 for expression of mouse Mesp1. A: ChIP analysis monitoring occupancy of TRF3, TBP, RNA polymerase II (POL2) or, as a negative control (C), an irrelevant protein (yeast Gal4) on the Mesp1 and Actin promoters in mouse E10.5 embryos. B: ChIP analysis monitoring promoter occupancy of TRF3, TBP, TAF3, and POL2 in mouse F9 embryonal teratocarcinoma cells. C: RT-PCR analysis of Trf3 and Mesp1 gene expression in F9 cells at 0, 1, 3, or 6 days after addition of retinoic acid (RA). D: RNAi. Trf3 double-stranded RNA (dsRNA) or, as a control, GFP dsRNA, was transfected into F9 cells and 48 hr later Trf3, Mesp1, Tbp, and Gapdh expression were monitored by RT-PCR analysis. E: Taf3 dsRNA or control (GFP) dsRNA was transfected into F9 cells and 48 hr later Taf3, Trf3, Mesp1, and Actin expression were analyzed by RT-PCR. |
|
Requirement of TRF3 for initiation of hematopoiesis in mouse embryoid bodies. A: RT-PCR analysis monitoring expression of various genes in mouse embryoid bodies at the indicated times (days) following induction of differentiation. B: Mouse embryonic stem cells were transfected with a Trf3 or control (luciferase) siRNA, differentiated into embryoid bodies, and 4 days later expression of various markers was analyzed by RT-PCR. |
|
Injection of the taf3 MO results in decreased levels of Taf3 in zebrafish embryos. Immunoblot analysis monitoring levels of Taf3 and tubulin in taf3 or control MO-treated embryos at 24 hpf. |
|
Expression of pu.1 is reduced in Taf3-depleted embryos. pu.1 expression was monitored in control and taf3 MO-treated embryos by RT-PCR analysis at 24 hpf. Expression of gapdh was monitored as a loading control. |
|
The developmental and hematopoietic defects observed in the Taf3-depleted embryos are not due to non-specific p53 activation. A: qRT-PCR analysis monitoring levels of p53, 113 p53 (an alternatively spliced isoform of p53 that is known to be highly upregulated in zebrafish following MO off-target effects) (Robu et al., 2007), p21 (a well-characterized p53 target gene), and gapdh in taf3 MO-treated embryos. As a control, expression of p53, 113 p53, and p21 were also monitored in control MO-treated embryos, which was arbitrarily set to 1. Error bars represent SD. The results show that the levels of p53, 113 p53 and p21 mRNAs are not significantly affected in Taf3-depleted embryos, indicating that the hematopoietic defect observed in the Taf3 morphants is not due to non-specific activation of the p53 pathway. B: Whole-mount in situ hybridization with a riboprobe to hbbe1 or gata1 (28 hpf) in embryos injected with a control MO or a taf3 MO, or co-injected with taf3 and p53 MOs. The results show that hbbe1 and gata1 expression in taf3 MO-injected embryos was equivalent to that obtained in embryos co-injected with taf3 and p53 MOs, indicating the hematopoietic defect was not due to p53 activation. C: Acridine orange staining of embryos (30 hpf) injected with a control MO or a taf3 MO, or co-injected with taf3 and p53 MOs. The results show that there was no difference in staining between Taf3 MO-injected embryos and those co-injected with MOs directed against both Taf3 and p53. |
|
Taf3 is expressed at a relatively constant level during differentiation in embryoid bodies. RT-PCR analysis monitoring expression of Taf3 in mouse embryoid bodies at the indicated times (days) following induction of differentiation. Expression of Hprt was monitored as a loading control. {FIGS6} |
|
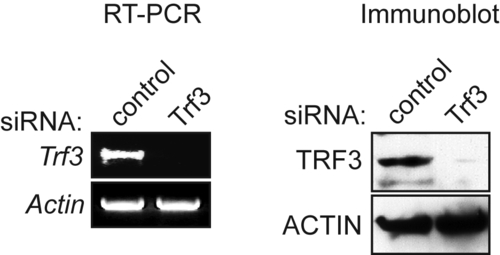
Analysis of Trf3 expression in mouse embryonic stem cells following siRNA-mediated knockdown of Trf3. RT-PCR (left) and immunoblot (right) analysis monitoring Trf3 knockdown following treatment with a control (luciferase) or Trf3 siRNA. Actin was monitored as a loading control. |