- Title
-
Zebrafish integrin-linked kinase is required in skeletal muscles for strengthening the integrin-ECM adhesion complex
- Authors
- Postel, R., Vakeel, P., Topczewski, J., Knöll, R., and Bakkers, J.
- Source
- Full text @ Dev. Biol.
|
Skeletal muscle detachments in loc/ilk mutant embryos. (A,B) Transmitted light images of tail regions in wt (A) and loc/ilk mutant embryos (B) at 4.5 dpf. Muscle fibre retractions are indicated with arrowheads. (C,D) Phalloidin staining and confocal images of wt sibling embryo (C) and of loc/ilk mutant embryo (D) at 4.5 dpf. Muscle fibre detachments are apparent in 50% of the loc/ilk mutant embryos (arrowheads). (E,F) α-actinin antibody staining of wt sibling (E) and loc/ilk mutant embryo (F) at 4.5 dpf. Regions of muscle fibre detachments are indicated by arrowheads. (G–J) Phalloidin staining and confocal images of uninjected control embryo (G), uninjected loc/ilk mutant embryo (H), dystroglycan MO injected wt embryo (I) and a dystroglycan MO injected loc/ilk mutant embryo (J). Tail regions of 2 dpf embryos are shown. |
|
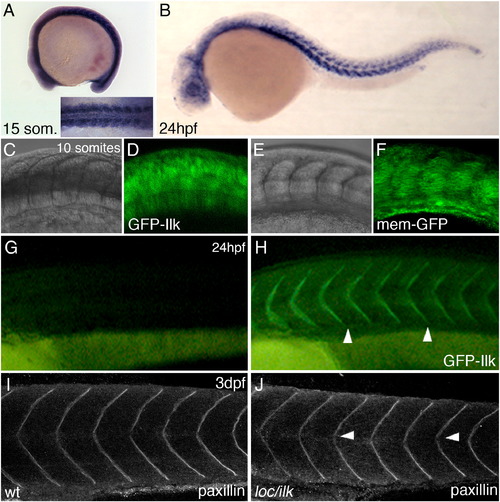
Ilk–GFP cellular localization. (A,B) Whole mount ISH with dig-labelled antisense ilk mRNA at 15-somite stage (A and inset) and 24 hpf (B). Wt embryos injected with synthetic mRNA encoding GFP–ILK (C and D) or memGFP (E and F). Images were taken at the 10-somite stage (15 hpf) at the region of the forming somites. The GFP–Ilk protein shows a predominant cytoplasmic localization at this stage. (G,H) Uninjected wt embryo (G) and a wt embryo injected with synthetic mRNA encoding GFP–Ilk (H) at 24 hpf. GFP–Ilk protein localization at the somite boundaries is indicated by arrowheads. (I,J) Anti-paxillin antibody staining and confocal images of a wt embryo (I) or loc/ilk mutant embryo (J) at 3 dpf. EXPRESSION / LABELING:
|
|
Knock-down of zebrafish itgα7 results in muscle fibre detachments. (A) Phylogenetic tree of the itgα6 and itgα7 sequence of several species. itgα1 of C. elegans and Drosophila was used as an outer group. (B,C) Whole mount ISH with dig-labelled antisense itgα7 mRNA at 15-somite stage (B) and 24 hpf with cross section (C). (D–F) Compared to wt embryos at 4 dpf (D) injections of itgα7-splice MO (E) or itgα7-ATG MO (F) result in muscle fibre detachments. EXPRESSION / LABELING:
PHENOTYPE:
|
|
GFP–Ilk and paxillin protein localizations in itgα7 and lama4 morphant embryos. (A–C) Injection of gfp–ilk mRNA in wt embryos results in GFP–Ilk protein localization to the MTJ at the somite boundaries in 24 hpf embryos (A). Reduced GFP–Ilk protein localization in embryos co-injected with an itgα7–ATG MO (B) or lama4–ATG MO (C). (D–F) Anti-paxillin antibody staining on wt embryos (D). Reduced paxillin localization to the MTJ at the somite boundary in itgα7–ATG MO (E) or lama4–ATG MO (F) injected embryos at 24 hpf. |
|
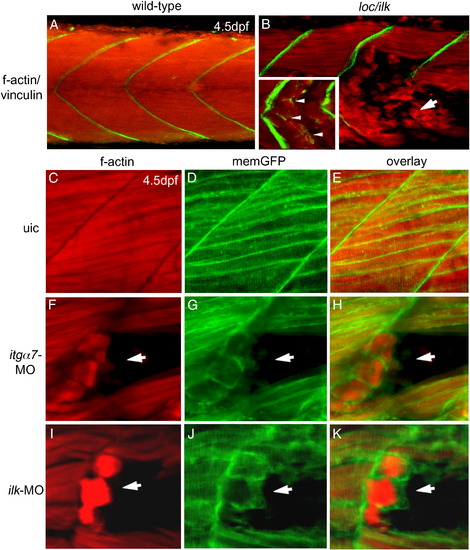
Plasma membrane retractions of skeletal muscle fibres. (A,B) Double labelling with anti-vinculin antibody (green) and phalloidin (red) in wt sibling embryos (A) and loc/ilk mutant embryos (B) at 4.5 dpf when muscle fibres detach (arrow). Inset shows vinculin staining at the tip of the f-actin filaments (inset in panel B; white arrowheads). (C–K) Double staining for phalloidin (red) and memGFP (green) as separate images or as an overlay taken from a wt embryo (C–E), an itgα7 MO injected embryo (F–H) and an ilk MO injected embryo (I–K) in a region where muscle fibres detached (arrow) at 4.5 dpf. |
|
Lysine 220 is essential for in vivo Ilk function. (A) Phosphorylation of Akt/PKB at ser 473 and GSK-3β at ser 9 is not altered in loc/ilk mutant embryos. Whole embryo lysates of wt sibling embryos and loc/ilk mutant embryos at 3 dpf show identical amounts of phosphorylated Akt/PKB and GSK-3β by Western blot analysis using an anti-phospho Akt/PKB ser 473 or an anti-phospho GSK-3β ser 9 antibody. Western blotting with a non-phospho specific Akt/PKB antibody using the same lysates shows identical levels of Akt/PKB protein present in both samples. Anti-GAPDH was used as an additional loading control. (B) Relative expression levels of Ilk in embryonic extracts of uninjected (control) or gfp–ilk, gfp–ilkK220M and gfp–ilkK220A mRNA injected embryos. (C–F) An uninjected control embryo with a weak and non-specific autofluorescence (C). Embryos injected with synthetic mRNA encoding wt GFP–Ilk (D), GFP–Ilk E359K (E) or GFP–Ilk K220M (F) all show a normal localization of the GFP–Ilk protein to the MTJs located at the somite boundary. (G–J) Muscle detachments are obvious in loc/ilk mutant embryos (G). The phenotypes including muscle detachments of loc/ilk mutant embryos were rescued by injection with synthetic mRNA encoding wt Ilk (H) or Ilk E359K (I). No rescue was observed in loc/ilk mutant embryos injected with mRNA encoding Ilk K220M (J). |
|
In vitro interaction of ILK with MLP and β-parvin. (A) Yeast two hybrid analysis of ILK interaction with Muscle lim protein (MLP) and β-parvin: Schematic diagram illustrates the recombinant expressed MLP and the full-length β-parvin. Below the human MLP are the full-length and the deletion mutants of mouse origin that were generated to assess the interaction between ILK and MLP. Schematic representations are as follows Control empty plasmid pGBKT7, hMLP (full-length), mMLP (full-length) and various regions of mMLP, and full-length β-parvin. Quantification of the interaction was based on beta-galactosidase activity with growth on medium (+) and stringent (++) selection media (see also Materials and methods section and Supplementary Table 1). (B–D) In situ hybridization with dig-labelled antisense mlp probe at 15 somite stage (B), 24 hpf (C) and 48 hpf (D). Arrowhead marks mlp mRNA expression in the heart. (E–G) In situ hybridization with dig-labelled antisense β-parvin probe at 12 somite stage (E), 30 hpf (F) and 48 hpf. Arrowheads marks β-parvin expression in the heart (G). (H,I) Phalloidin staining of f-actin in 5 dpf wt non-injected embryo (H) compared to mlp/crp3 MO injected embryo (I). No muscle fibre defects were observed in mlp/crp3 MO injected embryos. (J,K) Phalloidin staining of f-actin in 4 dpf wt non-injected embryos (J) compared to β-parvin-MO injected embryos (K). Severe muscle fibre detachments were observed in β-parvin morphants. EXPRESSION / LABELING:
|
|
Skeletal muscle differentiation in loc/ilk mutants. A-D) In situ hybridisation with dig-labelled antisense myoC (A,B) and myoD (C,D) probes at 24hpf on homozygous wt loc/ilk embryos (A,C) and mutant loc/ilk embryos (B,D). Both muscle differentiation markers are expressed normal in loc/ilk mutants. EXPRESSION / LABELING:
|
|
Morpholino mediated knockdown of dystroglycan. A,B) Phalloidin staining of f-actin on wt uninjected embryos (A) compared to dystroglycan-MO injected embryos at 4dpf (B). Severe muscle fibre detachments were observed in dystroglycan morphants. PHENOTYPE:
|
|
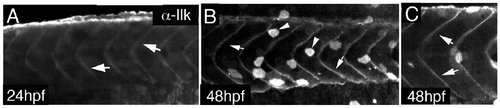
Ilk whole mount antibody staining. A-C) Whole mount antibody stain on wt embryos at 24hpf (A) and 48hpf (B,C). Ilk antibody localises at the somite boundaries at these stages. Non-specific staining in the skin is observed as well. |
|
Controls for morpholino mediated knock-down of itgα7 and mlp. A,C) RTpcr on cDNA of itgα7-splice MO (A) or mlp-splice MO (B) injected embryos shows severe reduction of the amount of wt transcript compared to uninjected control embryos (uic). As a control for the amount of cDNA, RT-PCR for ef1α was performed. Larger transcript are observed in both the itgα7- splice MO and mlp- splice MO injected embryos (asterisk). Sequencing of these larger PCR product confirmed the defective splicing of both transcripts resulting in a introduction of a premature stop codon. The arrowhead indicates an nonspecific PCR product. |
|
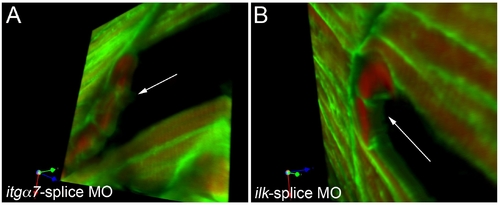
Morpholino mediated knockdown of itga7 or ilk causes retraction of the complete skeletal muscle cell. |

Unillustrated author statements PHENOTYPE:
|
Reprinted from Developmental Biology, 318(1), Postel, R., Vakeel, P., Topczewski, J., Knöll, R., and Bakkers, J., Zebrafish integrin-linked kinase is required in skeletal muscles for strengthening the integrin-ECM adhesion complex, 92-101, Copyright (2008) with permission from Elsevier. Full text @ Dev. Biol.