- Title
-
A role for the inositol kinase Ipk1 in ciliary beating and length maintenance
- Authors
- Sarmah, B., Winfrey, V.P., Olson, G.E., Appel, B., and Wente, S.R.
- Source
- Full text @ Proc. Natl. Acad. Sci. USA
|
ipk1 knockdown reduces normal ciliary beating and length. (A and B) Schematic diagrams (A) and kymographs (B) showing trajectory of a cilium in uninjected, ipk1 MO1-injected, ipk1 MO1 plus wild-type ipk1 mRNA-injected, or ipk1 MO1 plus kinase-dead (kd) ipk1 mRNA-injected embryos. (C) Graphs for measurements of ciliary length from individual embryos (green), with the mean ciliary length (far right, purple) determined from multiple independent measurements for KV (n > 26), pronephric duct (n > 13), and spinal canal (n > 14) cilia. (D) Representative images of KV cilia (red) from embryos injected with 10 ng of either control MO or ipk1 MO1. n, number of cilia measured per embryo. PHENOTYPE:
|
|
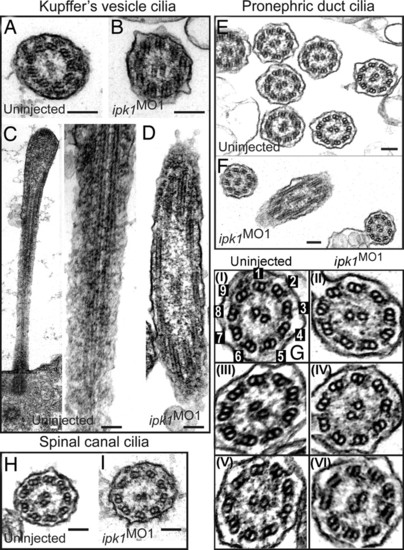
ipk1 knockdown does not affect ciliary axoneme organization. (A and B) TEM cross-sections of cilia in KV for uninjected (A) and ipk1 MO1 (B) embryos. (C and D) Longitudinal TEM sections showing axonemal microtubules in KV cilia in uninjected (C) and ipk1 MO1 (D) embryos. (E–I) TEM sections showing pronephric duct cilia (E, F, and G) and spinal canal cilia (H and I). Enlarged images of independent pronephric duct cilia TEM sections are shown in G. Cilia sections in uninjected (E, G-I, G-III, G-V, and H) and ipk1 MO1 (F, G-II, G-IV, G-VI, and I) embryos are shown. In G–I, numerical tags are assigned to each of the nine outer microtubule doublets for reference to dynein arms in the text. (Scale bars, 0.1 μm.) PHENOTYPE:
|
|
Ipk1 links to IFT and microtubule-mediated organelle transport. (A and B) Codepletion of Ipk1 and either IFT88 or IFT57 synergistically perturbs LR asymmetry. (A) Embryos were injected with varying amounts of either a single MO (ipk1 MO1, ift88 MO, ift57 MO, control MO) or a MO combination (ipk1 MO1+ift88 MO, ipk1 MO1+ift57 MO, control MO+ift88 MO, control MO+ift57 MO). (B) Embryos derived from crossing either heterozygous ift88 +/- mutant or wild-type fish were injected with ipk1 MO1 or control MO. Scores for heart-tube placement defect in embryos at 28 hpf are graphed. (C and D) ipk1 knockdown perturbs microtubule-mediated organelle transport. (C) Five days after fertilization embryos [uninjected and ipk1 MO1-injected (Left)] were treated with 0.5 mg/ml epinephrine, and time (min) required for all melanosomes in the head and trunk (within the white rectangle) to become perinuclear (Center) was determined. Time required for retracted melanosomes in epinephrine-treated endpoint embryos to fully disperse on exposure to 1 mg/ml caffeine (Right) was also determined. (D) Graph of the response time for epinephrine and caffeine treatments in uninjected and ipk1 MO1 embryos. |
|
Cilia (red) in pronephric ducts (Upper) and spinal canal (Lower) of embryos at 30 hpf injected with 10 ng of either control MO (Left) or ipk1MO1 (Right). Cilia were visualized by indirect immunofluorescence with anti-acetylated tubulin antibodies and laser scanning confocal microscopy. Arrowhead indicates cilia. PHENOTYPE:
|
|
Ipk1 depletion does not alter developmental stage-specific gene expression. Whole-mount in situ hybridizations are shown for embryos injected with 10 ng of either control MO (Left) and ipk1MO1 (Right). From top to bottom: Dorsal views of eight-somite-stage embryos showing krox20 expression in hindbrain rhombomeres 3 and 5 (Top), myoD in the somites (Middle), and deltaC in presomitic mesoderm and the posterior half of somites (Bottom). EXPRESSION / LABELING:
|
|
Codepletion of Ipk1 and either IFT88 or IFT57 synergistically perturbs LR asymmetry. Embryos obtained from crossing wild-type fish were injected with varying amounts of either single MO (ipk1MO1, ift88MO, ift57MO, control MO) or a MO combination (ipk1MO1 + ift88MO, ipk1MO1 + ift57MO, control MO + ift88MO, control MO + ift57MO). On the other hand, embryos derived from crossing heterozygous ift88 (previously known as polaris or oval) mutant (ift88+/-) fish were injected with varying amounts of either ipk1MO1 or control MO. Scores on the heart-tube placement defect in embryos at 28 hpf from each treatment are presented as bar graphs. PHENOTYPE:
|