- Title
-
Genetic determinants of hyaloid and retinal vasculature in zebrafish
- Authors
- Alvarez, Y., Cederlund, M.L., Cottell, D.C., Bill, B.R., Ekker, S.C., Torres-Vazquez, J., Weinstein, B.M., Hyde, D.R., Vihtelic, T.S., and Kennedy, B.N.
- Source
- Full text @ BMC Dev. Biol.
|
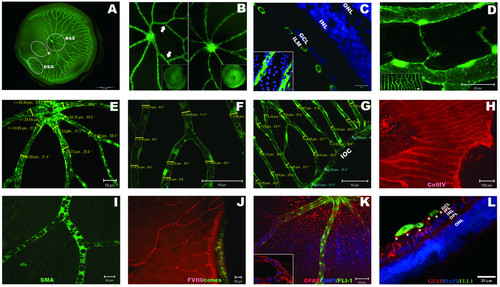
Adult zebrafish have a complex system of retinal blood vessels. A: Wholemount retina showing central major vessels that radiate into thinner vessels covering the entire inner surface of the retina. B: Example of disparity in branch number between left (5) and right (8) eyes. C: Transverse view of the blood vessels (green: Fli1-EGFP) overlaying the inner limiting membrane of the adult retina (blue: DAPI nuclear staining). Inset shows the same vessels overlying GCL nuclei in a flatmount preparation. D: At peripheral retinal regions, neighbouring vessels anastomose (inset), and elongated filopodia sprout from the capillaries suggesting active angiogenic remodelling. E-G: The diameter of vessels is thicker proximal to the optic disc and thinner peripherally. Numbers refer to the thickness of the vessel and the angle of measurement in reference to horizontal plane. H-K: Flatmount adult retinas immuno-labelled with retinal vasculature markers. H: collagen IV (red) stains the basal membrane of blood vessels. I: smooth muscle actin (SMA) stains vascular pericytes (green). J: Factor VIII labels endothelial cells (red). Cone photoreceptors label green as analysis was performed in Tg(3.2TαCP:EGFP) transgenic line [67]. K-L: Glial fibrillary acidic protein (GFAP) stains retinal vessels in adult zebrafish and a population of cells throughout the retina. GFAP (red), DAPI (blue) and fli1-EGFP (green). K: Flat-mounted retina, and L: Transverse view of the peripheral retina. Müller endfeet (asterisks) directly contact the endothelial cells (yellow co-staining). Inset in K: FITC channel turned off to highlight the GFAP reactivity of retinal vessels. ILM: inner limiting membrane; GCL: ganglion cell layer; IPL: inner plexiform layer; OPL: outer plexiform layer; INL: inner nuclear layer; ONL: outer nuclear layer; IOC: inner optic circle. EXPRESSION / LABELING:
|
|
Dynamic development of the hyaloid and retinal vasculature in zebrafish. Shown are fluorescent images of blood vessels on lenses and wholemount retinas dissected from Tg(fli1:EGFP) zebrafish. A: First intraocular vessels are detected at 60 hpf surrounding and attached to the forming lens. B: This vasculature evolves quickly and at 5 dpf covers the lens from the optic disk to the IOC. C: At 19 dpf an intricate network of hyaloid vessels branches around the lens. Some vessels at the posterior lens have lost contact and are attached to the retina (inset). D: At 28 dpf, detachment of the vessels from the lens has progressed anteriorly from the central region and extensive vasculature is found on the inner retina. E: Retina and F: lens from a 60 dpf zebrafish. In this specimen some vessels are attached to the lens although most of them are found on the retina. Inset in F: Overlay of the retina from E pseudo-coloured in red, and the lens from F depicts the complementing network of vessels. G: Retina of another 60 dpf zebrafish with the complete vasculature overlying the inner retina. H: Typical vascular pattern of intraocular vasculature in a 6 month old fish. I: Inner retina of a 22 month old senescent zebrafish showing slightly thinner and more fragile vessels. White circumferences demarcate the lens in A-F. Yellow arrows point from posterior to anterior lens in A-D&F and from dorsal to ventral retina in E&G-I. Scale bars: 50 μm in A-D; 100 μm in E-G and 500 μm in H-I. |
|
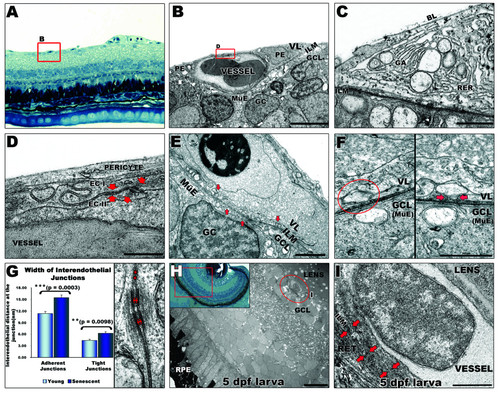
Ultrastructural analysis of the inner retina blood supply in zebrafish. A: Light microscopy image of blood vessels (highlighted by a red square) overlying the inner limiting membrane (ILM) of the retina in adult zebrafish. 40× magnification. B: Ultrastructure of capillary. The basal lamina (BL) is in contact with the ILM enclosing pericytes (PE) and the vascular endothelium. Scale bar: 5 μm. C: Typical structural features of pericytes, e. g. Golgi apparatus (GA), rough endoplasmic reticulum (RER) and large membrane bound vesicles. Scale bar: 1 μm. D: Vitreal space lined by pericyte overlying endothelial cells (EC) which display interdigitating junctional complexes (arrows). Scale bar: 500 nm. E: Multiple vesicles (arrows) from 20 to 250 nm contacting the inner limiting membrane of the retina indicate active interaction between the vessels and the ganglion cell layer (at the Müller endfeet). Scale bar: 1 μm. F: Vascular (top) and ganglion cell layer interface (bottom). A vesicle (left panel) apparently separated from the cell membrane, fuses with the cell membrane when the section is tilted by 43° (right panel) suggesting transcellular transport. Scale bar: 500 nm. G: Interendothelial junctions are significantly more open in the senescent fish. Distance between endothelial cells at tight (asterisks) and adherent (arrows) junctions were measured in different peripheral and central areas of retinas from senescent and young adult fish. Error bars: sem. Inset in H: Histology of a 5 dpf zebrafish eye. 40× magnification. H: EM image of a 5 dpf larval eye illustrating the relationship of hyaloid vessels (encircled by a red line) to both the lens and the retina. Scale bar: 10 μm. I: Higher magnification of the hyaloid vessel confirms its tight attachment to the lens and looser contact to the retina (arrows). Scale bar: 1 μm. MüE: Müller cell endfeet; GC: ganglion cell; GCL: ganglion cell layer; VL: vascular layer; RPE: retinal pigmented epithelium; RET: retina. |
|
Genes required for hyaloid and retinal vasculature development. A-E: Fluorescent images of fli1-EGFP hyaloid vessels on dissected lenses. A: Characteristic pattern of hyaloid vasculature attached to the lens of 5dpf wild type fish. B: Stagnated growth of hyaloid vasculature with less and thicker branches in MAGP1 morphants, and aggregation of vascular endothelial cells at the posterior lens. C: Aberrant oversized branches and lack of patterning in hyaloid vasculature of 5 dpf HS6ST2 morphants. D: hyaloid vessels in 5 dpf mab21l2 morphants are thicker and poorly patterned. E: Poor patterning and abnormal branching of the vessels attached to the lens of 5 dpf obd mutants F: Wholemount retina showing the retinal vasculature of an adult obd mutant exhibiting atypical loops (small arrows) and increased number of vessels. G-P: Alkaline phosphatase staining of hyaloid vasculature on lenses from mutant (left) and wild type larvae (right). G-H: Lens from mgf mutants is reduced in size and shows only residual unorganized vascular tissue at 4 dpf and no vessels at 5 dpf. I: Lens from fe mutants is slightly smaller and has no hyaloid vessels at 3 dpf. J-L: Lens from dsl mutants are much smaller but the hyaloid vasculature displays no apparent abnormalities at 5–8 dpf. M-O: Hyaloid vessels of plt mutants are loose at the back of the lens (asterisks) and the vascular pattern gradually becomes less intricate. P: Vasculature attached to lens of 4–6 dpf lop mutants is similar to vessels on wild type lens. Arrows point from posterior to anterior lens in all panels expect in 4F, where it points from dorsal to ventral retina. Scale bars: 50 μm in all panels except F, 500 μm. |
|
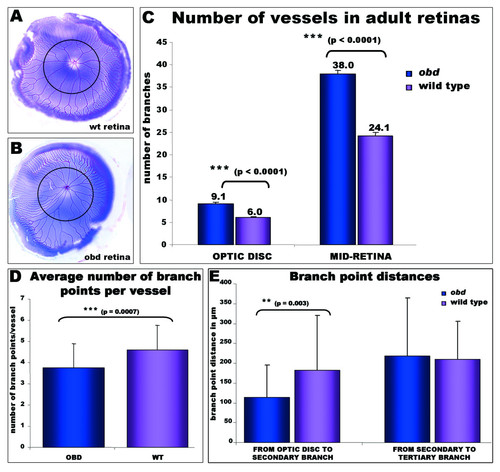
Adult-onset retinal vasculature phenotypes in Plexin D1 mutants. A-B: Pseudo-coloured adult retinas from (A) wild type and (B) obd adult fish to facilitate quantification of the vessel branches. A black circumferential line demarks half the distance between the optic disc and the annular peripheral vein at different retinal regions (MID- retina). C: The number of main branches radiating from the optic disc and the number of vessels crossing the mid-retina are higher in obd mutants (n = 18) than in wild type retinas (n = 24). D: The average number of branch points per vessel is significantly decreased in obd (n = 5) versus wildtype (n = 5) retinas. The number of branch points per 9 randomly chosen vessels in each retina was counted from outside in. To avoid bias, the nearest vessel was chosen every 40° of retinal circumference, starting from the most ventral poin. E: A significant reduction in the distance from the optic disc branches to the secondary branches, but not to the tertiary branches, is observed in obd vessels (n = 40) versus wildtypes (n = 22). PHENOTYPE:
|
|
Adult-onset retinal vasculature phenotypes in Plexin D1 mutants. A-B: Pseudo-coloured adult retinas from (A) wild type and (B) obd adult fish to facilitate quantification of the vessel branches. A black circumferential line demarks half the distance between the optic disc and the annular peripheral vein at different retinal regions (MID- retina). C: The number of main branches radiating from the optic disc and the number of vessels crossing the mid-retina are higher in obd mutants (n = 18) than in wild type retinas (n = 24). D: The average number of branch points per vessel is significantly decreased in obd (n = 5) versus wildtype (n = 5) retinas. The number of branch points per 9 randomly chosen vessels in each retina was counted from outside in. To avoid bias, the nearest vessel was chosen every 40° of retinal circumference, starting from the most ventral poin. E: A significant reduction in the distance from the optic disc branches to the secondary branches, but not to the tertiary branches, is observed in obd vessels (n = 40) versus wildtypes (n = 22). |
|
Physical interaction of Müller glia and retinal vasculature in adult zebrafish. A: Transverse view of peripheral retina in an adult Tg(gfap:EGFP) transgenic animal shows Müller cells (green) expanding through all retinal layers (blue: nuclear DAPI staining). B: Higher magnification shows Müller endfeet interposed with ganglion cell soma and contacting the retinal vessels (v). C: When the vascular layer is dissected from the inner interface of the retina (arrows) Müller endfeet remain attached to the vessels indicating a tight interaction. D: Blood vessel (green: Fli1-EGFP) overlying an adult retina seen from above with ganglion cell layer in the background (blue: DAPI nuclear staining). Müller cell endfeet (red: GFAP antibody) are observed on the entire surface of the inner retina, but especially concentrated along the retinal vessel, in direct contact with the vascular endothelium (yellow co-staining). GCL: ganglion cell layer; v: vessel; IPL: inner plexiform layer; INL: inner nuclear layer; OPL: outer plexiform layer; ONL: outer nuclear layer. EXPRESSION / LABELING:
|
|
Retinal vasculature in adult Plexin D1 mutants (obd) is characterised by a higher number of vessels and increased tortuosity. A: Left and right retinas from an obd mutant showing 9–10 main vascular branches radiating from the optic disc. Scale bar 1 mm. B: Higher magnification of an adult obd retina exhibiting extraneous vascular branches and loops that are never observed in wild types. Scale bar 200 μm. C: Plexin D1 obd mutant retina exhibiting increased vessel tortuosity. Scale bar 500 μm. OD: optic disc PHENOTYPE:
|