- Title
-
Induction and prepatterning of the zebrafish pectoral fin bud requires axial retinoic acid signaling
- Authors
- Gibert, Y., Gajewski, A., Meyer, A., and Begemann, G.
- Source
- Full text @ Development
|
Pectoral fin field induction requires retinoic acid signaling during somitogenesis. Anterior is towards the left: lateral (A-H,K-N), dorsal (I,J) and dorsolateral (O,P) views. Whole mount in situ hybridizations. (A-H) Wild-type embryos treated from 10 hpf onwards with 10-6 M and 5x10-6 M BMS493 (B,F and C,G) and with 10 µM DEAB(D,H); untreated controls (A,E). Expression of mesenchymal (tbx5) and apical epidermal fold (dlx2a) markers (arrows) is lost at the higher antagonist concentration and in DEAB treatments. (I,J) In the absence of RA synthesis from 5 hpf onwards (10 µM DEAB), tbx5 expression is restored to near-normal levels by treatment with 10-8 M RA from 11 hours onwards (arrows) (J). (K-N) 10-9 M RA (11-36 hpf) rescues fgf10 expression (K,L) and 5x10-9 M RA (11-36 hpf) rescues tbx15 expression (M,N) in nls. (O,P) wnt2b expression is not abolished in wild-type animals treated with DEAB from 11 hpf onwards. (Q) Schematic overview of the treatment applied to wild-type embryos depicted in A-J; shaded area depicts time window in which RA signaling is required. EXPRESSION / LABELING:
|
|
Reduction of somitic mesoderm in no tail/spadetail double-mutants. Whole-mount in situ hybridizations. Anterior is towards the left, dorsal (A-J) and lateral views (E'-J'). (A-D) pax2a expression confirms the presence of intermediate mesoderm in ntl/spt; glomeruli are compacted in ntl/sp (arrowheads). (E-H,E'-H') Expression of aldh1a2 in somites is largely unchanged in ntl (F,F'), but reduced in spt (G,G') and absent in ntl/spt double-mutant embryos (H,H'); expression in double-mutants is upregulated in the ventral trunk (arrows), consistent with expression in intermediate mesoderm. (I-J') Double-staining reveals aldh1a2 expression in the intermediate mesoderm (delimited by arrowheads) of wild-type and ntl/spt embryos; the latter lack myod expression (J,J'). EXPRESSION / LABELING:
|
|
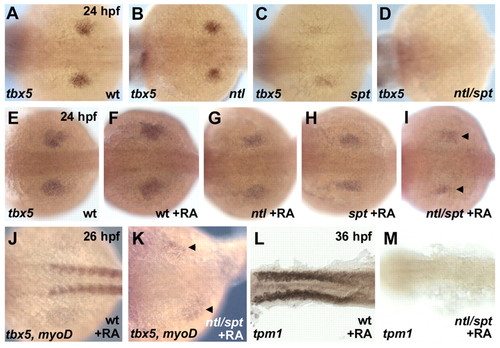
Retinoic acid rescues pectoral fin field induction in the absence of paraxial mesoderm. Whole-mount in situ hybridizations. Anterior is towards the left, dorsal views. (A-D) Expression of tbx5 in pectoral fin buds is unchanged in ntl(B), but reduced in spt (C) and absent in ntl/spt double-mutant embryos (D). (E-M) Application of 10-8 M RA, beginning at 10 hpf, rescues tbx5 expression in ntl/spt pectoral fins (arrowheads in I,K); rescue occurs in the absence of myogenic differentiation, demonstrated by the absence of myod (J,K) and α-tropomyosin-1 (tpm1) expression (L,M) in RA-treated double mutants. Four rescue experiments, each with 40 embryos, were performed (total numbers of phenotypes: 32 spt, 35 ntl, 11 ntl/spt, 82 siblings without mutant phenotype). |
|
Retinoic acid signaling from anterior somites rescues pectoral fin development in nls. Anterior is towards the left: lateral (A,B,D,E,G,H) and dorsal views (C,F). Transmitted light (A,C,D,F) and fluorescent (B,E,G,H) images taken at 48 hpf. (A-C) Wild-type donor cells (red fluorescence in B) occupying somites (s) 2-7 in a nls host are able to rescue pectoral fin (pec) development and tbx5 expression on the transplanted side (dark area in pectoral fin in A,B; arrow in C); chevron-shaped lines mark somite boundaries. The embryo in B was processed for in situ hybridization prior to photography, resulting in apparent enhancement of unspecific autofluorescence within the yolk. (D-F) Wild-type donor cells occupying most of somites 3-6 in a nls host rescue pectoral fin development (arrow in D) and tbx5 expression (arrow in F). (G,H) Donor cells transplanted to the dorsal aspect of somites 1-7 (G) or to somites posterior to somite 6 (H) are unable to rescue pectoral fin development (not shown). Scale bar: 100 µm. |
|
Expression of aldh1a2 in wild-type pectoral fins. Whole-mount in situ hybridizations. Anterior is towards the left: dorsal (A) and lateral views (B-F). Pectoral fin buds at 28 hpf (A,B), 32 hpf (C), and 2, 3 and 5 dpf (D-F). (A,B) aldh1a2 is expressed in the posterior part of developing pectoral fins and adjoining lateral plate mesoderm; bracket demarcates the fin proper. (C,D) aldh1a2 expression is restricted to the fin mesenchyme. (E,F) From 3 dpf onwards, expression is restricted to the proximal, anterior and distal margins of the mesenchyme, and is downregulated in the distal margin by 5 dpf. Scale bars: 100 µm. |
|
Expression of marker genes in the mesenchyme and ectoderm of wild-type and pectoral fins. Treatment with 10 µM DEAB, starting from 16 hpf to 28 hpf (A-D), 38 hpf (I,J) and 40 hpf (E-H,K,L); and from differing starting times to 32 hpf (M-P). Anterior is towards the left. (A,B) hand2 is expressed in the medial and posterior mesenchyme of wild type (arrow), but is not expressed in the absence of RA. (C,D) shh is expressed in the posterior mesenchyme (arrow), but is not expressed in the absence of RA. (E-H) hoxd11 and hoxd12 are expressed in the posterior mesenchyme and fail to be induced in the absence of RA. (I,J) hoxc6, which is normally restricted to the anterior mesenchyme, is expanded posteriorly in DEAB-treated embryos. (K,L) dlx2a, a marker of the apical fold, is normally expressed in the absence of RA. (M-P) shh induction is lost upon early inhibition of RA signaling. Scale bars: 50 µm. |
|
RA requirement in pectoral fins after 28 hpf. (A-C) Alcian Blue stains of larval pectoral fin and girdle at 6 (A,B) and 4,5 dpf (C). Treatment with 10 µM DEAB from 10-17 hpf inhibits formation of the scapulocoracoid (sco) (B), and from 28-50 hpf affects differentiation in the proximal skeletal elements of the pectoral girdle (C). (D-G) In situ hybridizations of pectoral fins at 50 hpf; differentiation of the fin mesenchyme (D,E), appendicular muscles (F) and apical fold (G) appear unchanged upon treatment with 10 µM DEAB. cl, cleithrum; ed, endoskeletal disc. EXPRESSION / LABELING:
|