- Title
-
The zebrafish kohtalo/trap230 gene is required for the development of the brain, neural crest, and pronephric kidney
- Authors
- Hong, S.K., Haldin, C.E., Lawson, N.D., Weinstein, B.M., Dawid, I.B., and Hukriede, N.A.
- Source
- Full text @ Proc. Natl. Acad. Sci. USA
|
Positional cloning of the zebrafish kto locus. (A) Genetic mapping of the kto locus on chromosome 14, not precisely to scale. Below the cM scale, markers that are polymorphic in a map cross with the TL strain are shown in black, and markers polymorphic in the WIK map cross are shown in red. Recombinants per number of meioses tested are listed below. Note that we have repositioned simple sequence length polymorphism marker z22128
|
|
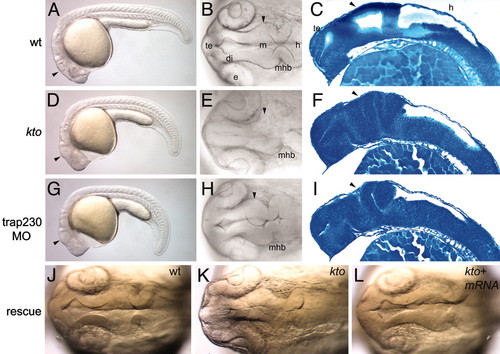
Brain phenotype of kto mutants. Embryos at 24 hpf. Live embryos (A, B, D, E, G, H, J-L) and sagittal sections (C, F, and I) stained with methylene blue. Lateral views (A, D, and G) and dorsal views (B, E, H, J-L) of the brain. Wild type (A-C), kto mutant (D-F), and embryos (G-I) injected with 2 ng of trap230 MO. Rescue of mutant (J-L), wild type (J), and kto embryo (K and L) injected with 100 pg of trap230 mRNA; the embryo was genotyped to ascertain that it was mutant. Arrowheads point to regions malformed in mutants and morphants. di, diencephalon; e, eye; m, midbrain; mhb, mid-hindbrain boundary; te, telencephalon.
|
|
The brain does not extend properly in kto mutants. Wild-type (A and C-E) and kto mutant (B and F). (A-D) In situ hybridization with flh; a gap (arrowhead) is seen in expression between telencephalon and diencephalon (epiphysis) in wild-type but not in kto embryos (B) or embryos injected with 2 ng of trap230 MO (D). (E and F) wnt1 expression in dorsal midbrain and midhindbrain boundary. Arrow shows reduced midbrain size in kto mutants. di, diencephalon; m, midbrain; mhb, mid-hindbrain boundary; te, telencephalon.
EXPRESSION / LABELING:
|
|
Neural crest development in kto embryos. Wild type (A, C, and E) and kto (B, D, and F) at the six-somite stage. snail2 expression (A and B) and sox9b expression (C and D) are reduced in the mutant. (E and F) Cell death as visualized by TUNEL analysis. The yolk has been removed. A moderate increase in dying cells can be seen in the mutant. e, eye; h, hindbrain; ov, otic vesicle; sp, spinal cord; tb, tail bud.
EXPRESSION / LABELING:
|
|
Reduced pigmentation in kto mutant embryos. Lateral views. (A and B) Pigment patterning of wild-type and kto embryos at 48 hpf. Pigmentation is reduced in the mutant embryo, especially in ventral regions. (C and D) nacre/mitfa expression at 24 hpf. Arrows and arrowheads point to dorsal neural tube and migrating neural crest cells, respectively. n, notochord.
EXPRESSION / LABELING:
|
|
Defects in the segmentation of pharyngeal arches in kto mutants. (A-D, I, J, O, and P) Lateral views, all others are ventral views. (A and B) Dlx2a expression in pharyngeal arches at 26 hpf. No distinct pharyngeal arches form (arrow), and expression in posterior arches is essentially extinguished in kto mutants (arrowhead). (C and D) TG(fli1:EGFP)y1 transgenic embryos at 31 hpf. In kto mutant embryos, p1 and p2 seem malformed and fused, whereas the more posterior arches are almost entirely lost (arrowhead). (E and F) dHAND expression in pharyngeal arches at 36 hpf. Arrows indicate separation of p1 and p2. In kto embryos, separation of p1 and p2 failed, and p3-p7 are largely absent. dHAND expression in the heart is present (white asterisk), but the pectoral fins are lost in kto.(G and H) Expression of nkx2.3 at 36 hpf visualized endodermal pouches; cells separating individual arches are lost in the mutant (arrowhead). (I and J) zn5 stains the endodermal pouches, trigeminal ganglia, and sensory neurons. (K and L) Alcian blue staining at 5 dpf. No cartilage elements are visible in the mutant. (M and N) Detection of larval muscles with F59 antibody at 4 dpf; muscles in the head are lost in the mutant. (O-R) Calcein-stained fluorescent image shows loss of bones in the head of kto embryos. b, branchyal arch; bh, basihyal; cb, ceratobranchial; ch, ceratohyal; e, eye; fb, forebrain; h, hyoid arch; hh, hyohyoideus; hs, hyosymplectic; ht, heart; ih, interhyoideus; imp, intermandibularis posterior; m, mandibular arch; mc, Meckel′s cartilage; n, notochord; ov, otic vesicle; p, pharyngeal arch; pf, pectoral fin, r, rhombomere; sh, sternohyoideus; te, teeth; tg, trigeminal ganglia; tv, transversus ventralis.
EXPRESSION / LABELING:
|
|
Anterior nephrogenic mesenchyme migration fails in kto embryos. Wild-type (A, C, E, and F) and kto mutant (B and D); at the five-somite stage, mutant embryos were identified by genotyping. (A and B) Five-somite stage, (C-F) 24 hpf. Dorsal views of wt1 expression show that the glomerulus does not coalesce at the midline in kto (D) and trap230 MO-injected embryos (F).
EXPRESSION / LABELING:
|