- Title
-
Zebrafish deadly seven functions in neurogenesis
- Authors
- Gray, M., Moens, C.B., Amacher, S.L., Eisen, J.S., and Beattie, C.E.
- Source
- Full text @ Dev. Biol.
|
Somite boundary formation is aberrant in desb420 mutants. Lateral views of live 18-h wild-type (A–C) and mutant (D–F) embryos at the level of anterior trunk (segments 2–5; A, D), mid-trunk (segments 8–12; B, E), and tail (segments 14–18; C, F). Black arrowheads denote normal somite boundaries and white arrowheads denote incomplete somite boundaries. In this and all subsequent lateral views, anterior is to the left and dorsal is to the top. Scale bar, 25 μm. PHENOTYPE:
|
|
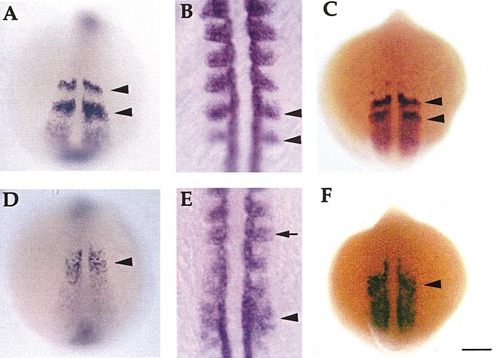
Somite and myotome gene expression is perturbed in desb420 mutants. Dorsal views (anterior to the top) of whole-mount RNA in situ hybridization of her1 (A, D; 13 h), myoD (B, E; 15 h), and mespa (C, F; 13 h) in wild-type (A–C) and mutant (D–F) embryos. Black arrowheads denote bands of gene expression in wild-type embryos and the corresponding region in mutants. Arrow in (E) designates normal myoD expression in anterior somites. Scale bar, 150 μm (A, C, D, F), 50 μm (B, E). PHENOTYPE:
|
|
Motor nerves are disorganized in desb420 mutants. Lateral views of whole-mount embryos labeled with the znp1 monoclonal antibody at 24 (A, C) and 48 h (B, D) in wild-type (A, B) and mutant (C, D) embryos. Arrowheads point to one axon (A, C) or nerve (B, D). Scale bar, 50 μm. |
|
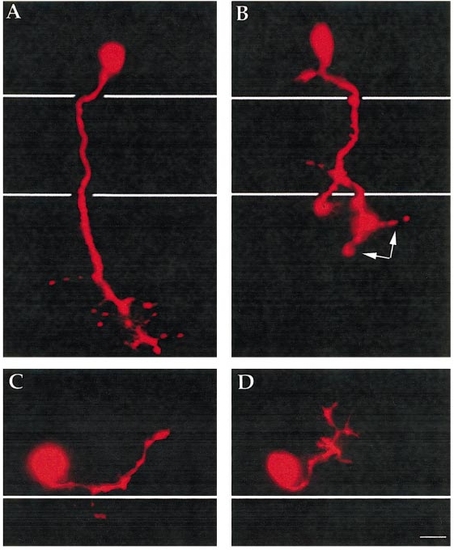
CaP and MiP motor axons display abnormalities in desb420 mutants. Lateral view of pseudocolor images of live CaP (A, B) and MiP (C, D) motoneurons labeled with rhodamine dextran and visualized at approximately 26 h in wild-type (A, C) and desb420 mutant (B, D) embryos. Cell bodies reside in the spinal cord and white lines denote the dorsal and ventral aspects of the notochord (A, B) and the dorsal aspect of the notochord (C, D). Scale bar, 10 μm. PHENOTYPE:
|
|
desb420 is non-cell autonomous for CaP motoneurons. Single CaP motoneurons were removed from rhodamine dextran labeled donors at approximately 16 h, a time before axogenesis, and transplanted into stage-matched, unlabeled host embryos from which native CaP and VaP motoneurons had been removed. (A) Wild-type CaP transplanted into wild-type host (n = 4). (B) Wild-type CaP transplanted into a desb420 mutant host (n = 4). (C) desb420 mutant CaP transplanted into wild-type host (n = 4). White lines denote dorsal and ventral aspects of the notochord. Scale bar, 10 μm. |
|
Migrating neural crest and neural crest derivatives are differentially affected in desb420 mutants. Lateral view of mid-trunk (segments 9–14) neural crest on the medial pathway as revealed by crestin expression (arrowheads) at 24 h in wild-type (A) and mutant (B) embryos. Cross section view of Hu-positive DRG neurons (arrowheads) in 72-h wild-type (C) and mutant (D) embryos. Lateral view of mid-trunk (segments 9–14) dct-expressing cells in wild-type (E) and mutant (F) embryos along the medial pathway. Pigment cells positioned in the lateral stripe in 3 d wild-type (G) and mutant (H) embryos. Scale bar, 50 μm (A, B); 30 μm (C–F); 75 μm (G, H). PHENOTYPE:
|
|
desb420 mutants have excess primary motoneurons. CaP and VaP motoneurons (arrowheads) as revealed by islet2 RNA in situ hybridization at 20 h in a wild-type (A) and mutant (B) embryo. (C) Counts of islet1 and islet2 expressing cells at 20 h in spinal hemisegments 5–9; n = 10 embryos for each point. For these and all subsequent histograms the mean ± 95% confidence interval is plotted. Significance was determined by Student’s t test with *, P = 0.01–0.001; **, P < 0.001. Scale bar, 25 μm. PHENOTYPE:
|
|
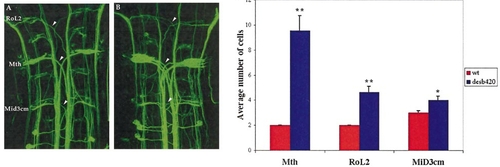
Supernumerary reticulospinal neurons are present in desb420 mutants. Dorsal view of a confocal image (anterior to the top) of a RMO44 labeled wild-type (A) and desb420 mutant (B) embryo. Somata of affected cells are labeled in (A) and arrowheads point to their corresponding axons. Note the increased number of axons in the mutant embryo. (C) Cell counts of affected reticulospinal neurons. Cells on both sides of the midline were analyzed for wild-type (n = 14) and mutant (n = 14) embryos. See Fig. 7 legend for details regarding statistics. Mth, Mauthner cell. Scale bar, 20 μm. PHENOTYPE:
|
|
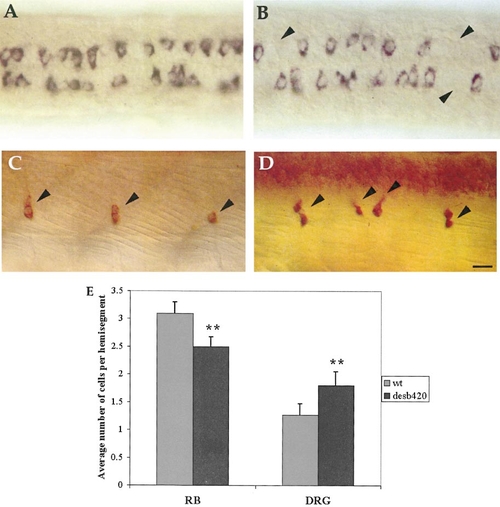
Analysis of trunk sensory neurons in desb420 mutants. Dorsal view (anterior to the left) of a 20-h wild-type (A) and mutant (B) embryo showing islet2 expressing RB neurons. Arrowheads denote the absence of RB neurons in the spinal cord of desb420 mutants. All three des alleles examined had a similar decrease in RB cells (b420 = 2.5 ± 0.2, b638 = 2.6 ± 0.2, and tp37 = 2.3 ± 0.1 compared to wild type = 3.1 ± 0.2). Lateral view of 72-h wild-type (C) and mutant (D) embryo showing Hu-expressing DRG neurons (arrowheads). (E) islet2-expressing RB neurons and Hu-expressing DRG neurons were counted in hemisegments 5–9; n = 10 embryos for RB counts and n = 6 embryos for DRG counts. See Fig. 7 legend for details regarding statistics. Scale bar, 20 μm. PHENOTYPE:
|
|
Notch rescues the desb420 Mauthner cell defect. Embryos from wild-type or heterozygous desb420 matings were injected at the 1- to 2-cell stage with RNA encoding Notch-ICD-myc (Coffman et al., 1993; Chitnis et al., 1995). Embryos were fixed at 28 h and processed for 3A10 antibody labeling to visualize Mauthner cells. (A) Mock-injected wild-type embryos have one Mauthner cell on either side of hindbrain rhombomere 4. (B) Approximately 25% of mock-injected embryos from a heterozygous desb420 mating have supernumerary Mauthner cells. Injecting RNA encoding Notch-ICD decreases the number of Mauthner cells in wild-type (C) and desb420 mutant (D) embryos. |

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. |

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. PHENOTYPE:
|
Reprinted from Developmental Biology, 237(2), Gray, M., Moens, C.B., Amacher, S.L., Eisen, J.S., and Beattie, C.E., Zebrafish deadly seven functions in neurogenesis, 306-323, Copyright (2001) with permission from Elsevier. Full text @ Dev. Biol.